Heat capacity is a crucial concept in thermodynamics that measures the amount of heat energy required to raise the temperature of a substance by one degree Celsius. It’s essential for IIT JAM and CSIR NET aspirants to understand Thermal Capacity, its units, and its applications.
Thermodynamics Syllabus and Key Textbooks for IIT JAM
If you’re diving into the IIT JAM thermodynamics syllabus, you’ve probably noticed that thermodynamics is a massive pillar. Specifically, chapters 1 through 3 are where the magic happens—this is where you get comfortable with systems, state functions, and, of course, heat capacity.
Think of thermodynamics as the “engine room” of your prep. If you don’t get these basics down, the more complex topics later on will feel like a struggle. To get a solid grip on this, most of us at VedPrep swear by textbooks like Halliday-Resnick-Walker or Cengel. They don’t just throw math at you; they actually explain the “why” behind the energy changes. Mastering heat capacity now will give you a huge head start for the 2027 exam cycle.
Heat Capacity: A Fundamental Concept in Thermodynamics for IIT JAM
So, what exactly is heat capacity? In simple terms, it’s just a measure of how much “heat hunger” a substance has. It’s the amount of energy you need to dump into something to raise its temperature by one degree.
Because it’s an extensive property, size matters. A bucket of water has a much higher thermal capacity than a cup of coffee, even if they’re at the same temperature. In the exam, you’ll usually see this expressed in Joules per Kelvin (J/K).
You’ll also run into two cousins: specific heat capacity (how much heat 1 gram needs) and molar heat capacity (how much heat 1 mole needs).
-
Specific: Think of it like the “unit price” per gram.
-
Molar: The “unit price” per mole.
At VedPrep, we often see students get these mixed up in a rush, so always double-check your units (J/g· K vs J/mol · K) before you bubble in that answer.
Worked Example: Calculating Heat Capacity For IIT JAM
Let’s look at a quick problem. Say you have 2 moles of CO2 being heated in a sealed, rigid container (constant volume). The molar heat capacity (CV,m) is 40.8\text{ J/mol · K. What’s the total capacity for this specific sample?
Since the volume isn’t changing, we use the formula:
Plugging in the numbers:
Common Misconceptions about Heat Capacity in IIT JAM Preparation
A common trap aspirants fall into is thinking heat capacity only matters when things are boiling or freezing. That’s actually a myth. It matters across the board—even near absolute zero.
Imagine a fictional scenario where you’re researching materials for a super-cooled quantum computer. At those bone-chilling temperatures, the way atoms vibrate changes completely. You can’t just use a “standard” number you found for room temperature. This is where models like the Debye model come in.
The big takeaway? Heat capacity isn’t a static number; it changes depending on the temperature. Some materials get “hungrier” for heat as they get warmer, while others don’t. Keep an eye on those temperature-dependent graphs in your practice sets. Thermal Capacity For IIT JAM aspirants should be aware that heat storage capacity can exhibit different behaviors, such as increasing or decreasing with temperature, depending on the specific substance and conditions.
Real-World Applications of Heat Capacity in IIT JAM
Why do we care about this outside of a lab? Well, it’s the reason your fridge works. Refrigeration systems move heat around using fluids with very specific thermal properties. If the heat exchanger’s capacity is off, your milk spoils, and your electricity bill skyrockets.
It’s also why coastal cities have milder weather. Water has a massive heat capacity. It absorbs a ton of sun during the day without getting too hot and releases it slowly at night.
In engineering, we use this to store energy. Imagine a fictional “solar salt” battery that stores heat during the day to keep a city running at night. The efficiency of that whole setup depends entirely on the heat capacity of the salt. Understanding these real-world vibes makes the abstract math a lot easier to swallow.
Exam Strategy: Tips for Solving Heat Capacity Problems in IIT JAM
When you’re staring down a thermodynamics question, don’t panic. Start with the most basic relationship:
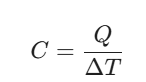
Here’s a quick game plan we suggest at VedPrep :
-
Check the constraints: Is the volume constant (CV) or is the pressure constant (CP)?
-
Watch the “Per”: Are they asking for specific (per gram) or molar (per mole)?
-
Practice the tricky stuff: The relationship between CP and CV (like CV – CP = R for ideal gases) is a favorite for paper setters.
By following these tips and utilizing resources like VedPrep, students can improve their chances of success in IIT JAM and other competitive exams, such as CSIR NET and GATE.
Heat Capacity for IIT JAM: Key Concepts and Formulas
To accurately solve thermodynamics problems for the IIT JAM examination, it is essential to master the various expressions of heat capacity. Below are the key formulas you will encounter:
-
General Heat Capacity (C):
Defined as the amount of heat (Q) required to produce a temperature change (ΔT):
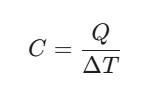
Unit: J/K or J/°C
- Specific Heat Capacity (c):
The heat capacity per unit mass (m) of a substance:
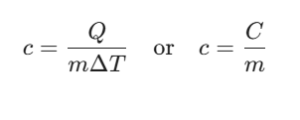
Unit: J/(kg·K) or J/(g·K)
- Molar Heat Capacity (Cm):
The heat capacity per mole (n) of a substance:
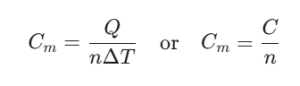
Unit: J/(mol·K)
Conclusion
At the end of the day, mastering heat capacity isn’t just about passing a test. It’s about understanding how energy moves through the universe. Remember, consistency is your greatest asset. Whether you are navigating complex molar thermal capacity calculations or applying these principles to materials science, every practice problem brings you closer to your goal. Stay curious, keep solving, and leverage the comprehensive resources at VedPrep to turn these challenging concepts into your strongest assets.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
What is the SI unit of heat capacity?
The SI unit for heat capacity is Joules per Kelvin (J/K) or Joules per degree Celsius (J/°C).
Is heat capacity an extensive or intensive property?
Heat capacity is an extensive property because its value depends on the total quantity of matter (mass or moles) present in the system.
What is the difference between specific heat and molar heat capacity?
Specific heat capacity is defined per unit mass (J/g-K), whereas molar heat capacity is defined per unit mole (J/mol-K).
Does heat capacity change with temperature?
Yes, for most substances, heat capacity is temperature-dependent. This is especially significant at low temperatures where quantum effects, such as those described by the Debye model, come into play.
What is the fundamental formula for heat capacity?
The basic formula is C = Q/ΔT, where Q is the heat exchanged and ΔT is the change in temperature.
How important is heat capacity for the IIT JAM Physical Sciences syllabus?
It is a high-yield topic. Understanding heat capacity is essential for mastering the First and Second Laws of Thermodynamics, which frequently appear in the exam.
Which chapters in the IIT JAM syllabus cover heat capacity?
Heat capacity is typically covered within the "Thermodynamics" unit, often appearing in chapters related to internal energy, enthalpy, and phase transitions.
What types of numerical problems on heat capacity appear in IIT JAM?
Common problems include calculating heat required for phase changes, determining internal energy changes, and applying Mayer's relation to ideal gases.
Are there common misconceptions about heat capacity?
Yes, students often mistakenly believe it is only relevant for high-temperature industrial processes, ignoring its critical role in low-temperature physics and statistical mechanics.
How can I master heat capacity problems for IIT JAM?
Mastery comes from consistent practice. Start by memorizing core definitions, then solve previous year's questions (PYQs) and utilize VedPrep’s expert study resources for guided practice.
What is the role of the Debye model in heat capacity?
The Debye model explains the heat capacity of solids at low temperatures by accounting for the quantization of lattice vibrations (phonons).
How is heat capacity used in refrigeration systems?
In refrigeration, heat capacity determines how efficiently heat exchangers can transfer thermal energy to maintain cooling cycles.
Is heat capacity relevant to materials science?
Absolutely. Engineers use heat capacity data to design thermal energy storage, heat sinks, and thermal protection systems in materials science.
Where can I find more practice problems on heat capacity for IIT JAM 2027?
VedPrep provides an extensive library of practice problems, mock tests, and detailed study modules tailored specifically for the IIT JAM 2027 curriculum.










