Equilibrium constant (Kp, Kc, Kx) For IIT JAM is a crucial concept in physical chemistry that describes the equilibrium between reactants and products in a chemical reaction, with Kp and Kc representing different units of measurement.
Chemical Equilibrium Syllabus for IIT JAM, CSIR NET, and GATE
If you open up Chapter 2 of the IIT JAM Physical Chemistry syllabus, you’ll find chemical equilibrium sitting right there. It’s a heavyweight topic. If you love flipping through classic textbooks, you’ve probably seen this broken down beautifully in Physical Chemistry by Atkins and De Paula, or McQuarrie and Simon’s Physical Chemistry: A Molecular Approach.
These books do a great job with the deep theory, but when you’re in the exam hall, you need to know how to apply that theory fast. That’s why we at VedPrep love breaking these down into actionable shortcuts. At its core, the equilibrium constant is just a ratio that tells you if you’ve got more products or reactants when the reaction finally settles down.
Understanding Equilibrium Constant (Kp, Kc, Kx) For IIT JAM
Imagine a crowded coffee shop. People are walking in, and people are leaving. Equilibrium isn’t when the inside of the shop looks empty; it’s when the rate of people entering perfectly matches the rate of people walking out. The total number of people inside stays constant.
In chemistry, the equilibrium constant tracks this balance, but we use different versions depending on what state of matter we are dealing with:
-
Kc (Concentration): This is your go-to for aqueous solutions. We measure the species in molarity (moles per liter).
-
Kp (Partial Pressure): Used when gases are playing the main role. Instead of molarity, we track the individual pressure each gas exerts.
-
Kx (Mole Fraction): This looks at the ratio of moles of one component to the total moles. It’s incredibly handy for mixtures and complex systems.
A quick heads-up: while some older texts casually call these “dissociation constants,” they are all specific types of equilibrium constants. The one you choose simply depends on the units you have on hand.
Worked Example: Equilibrium Constant (Kp, Kc, Kx) For IIT JAM
Let’s look at a classic problem you might encounter on exam day:
Imagine this reaction is trapped in a closed container at a total pressure of 100 bar. After things settle down, a sensor tells us the mole fractions (xi) for CO, H2, and CH3OH are 0.2, 0.4, and 0.4.
To find Kp, we need partial pressures first. We get these by multiplying each mole fraction by the total pressure (pi = xi × Ptotal:
-
pCO = 0.2 × 100 = 20 bar
-
pH2 = 0.4 × 100 = 40 bar
-
pCH3OH = 0.4 × 100 = 40 bar
Now, write out the expression for Kp. Remember, products go on top, reactants on the bottom, and coefficients become exponents:
Plug in our numbers:
![]()
Application of Equilibrium Constant (Kp, Kc, Kx) For IIT JAM in Real-World Scenarios
To make this feel less like abstract math, let’s look at a fictional scenario. Imagine a chemical engineer named Sarah who runs a massive industrial plant trying to make ammonia via the Haber-Bosch process:
If Sarah blindly mixes the gases without checking Kp, she might end up wasting thousands of dollars producing almost nothing. By using the equilibrium constant, she knows exactly how much pressure to apply to force the reaction to favor ammonia.
The same logic applies when creating methanol from carbon monoxide and hydrogen. Companies use Kc to figure out the sweet spot for temperature and pressure so they don’t waste energy. From developing new pharmaceuticals to designing wastewater treatment filters, these constants give engineers a predictable blueprint of what a chemical system will do before they even turn on the machines.
Exam Strategy: Equilibrium Constant (Kp, Kc, Kx) For IIT JAM
When you are prepping for IIT JAM, you can’t just memorize definitions. You need a game plan. The examiners love to test how you handle shifts and changes.
Here are the absolute essentials you need to focus on:
-
Units matter: Pay close attention to whether the question asks for bar, atm, or mol/L.
-
The Interconversions: You must know how to switch between constants effortlessly.
-
Le Chatelier’s Principle: Understand how changing temperature or pressure shifts the equilibrium position, and more importantly, how temperature is the only thing that actually changes the value of Kp and Kc.
At VedPrep, we always tell students that practicing a wide variety of problems is the only way to build true speed. Working through mock tests helps you spot the tricks examiners hide in the question text before they catch you off guard.
Important Equations and Formulas for Equilibrium Constant (Kp, Kc, Kx) For IIT JAM
Let’s lay out the mathematical tools you need. For a standard hypothetical reaction:
![]()
Here are the formulas you should commit to memory:
1. The Core Expressions
-
For Concentration:
- For Partial Pressure:
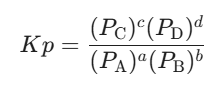
- For Mole Fraction:
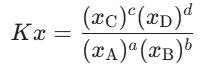
2. The Bridge Equations
The real magic happens when you need to link these together.
-
Connecting Kp and Kc:
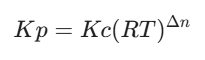
- Connecting Kx and Kc:
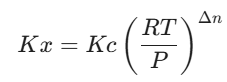
Note on Δn: This is just the number of moles of gaseous products minus the moles of gaseous reactants (Δn = (c+d) – (a+b)). If Δn = 0, then Kp = Kc = Kx, which is a favorite shortcut for exam setters!
Final Thoughts
Mastering the equilibrium constant for IIT JAM is less about mindless memorization and more about training your brain to think with analytical precision. Once you see how Kp, Kc, and Kx connect, the problems start solving themselves.
We know how overwhelming exam prep can feel when you are balancing multiple subjects. That’s exactly why we create targeted study materials and specific mock tests here at VedPrep—to help take the guesswork out of your routine. Keep working through the problems and get comfortable with the unit conversions, and you’ll find yourself cruising through the physical chemistry section with confidence.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
What are Kp, Kc, and Kx?
Kp, Kc, and Kx are equilibrium constants expressed in terms of partial pressures, concentrations, and mole fractions, respectively.
How is Kc related to Kp?
Kp and Kc are related by the equation Kp = Kc(RT)Δn, where Δn is the difference in the number of moles of gas between products and reactants.
What is the significance of Kx?
Kx is the equilibrium constant expressed in terms of mole fractions, which is useful for reactions involving gases or non-ideal solutions.
How is equilibrium constant related to reaction quotient?
The reaction quotient (Q) is the ratio of product concentrations to reactant concentrations at any point in the reaction, while the equilibrium constant (K) is the value of Q at equilibrium.
What are the units of equilibrium constant?
The units of Kc depend on the reaction, while Kp is unitless. Kx is also unitless.
Can equilibrium constant be negative?
No, the equilibrium constant is always positive.
How to calculate Kp from Kc?
To calculate Kp from Kc, use the equation Kp = Kc(RT)Δn and ensure that the units are consistent.
What are the applications of equilibrium constant in Physical Chemistry?
The equilibrium constant has numerous applications in Physical Chemistry, including predicting reaction spontaneity and calculating concentrations at equilibrium.
How to determine the equilibrium constant experimentally?
The equilibrium constant can be determined experimentally by measuring the concentrations of reactants and products at equilibrium and then calculating K.
What are the factors affecting equilibrium constant?
The equilibrium constant is affected by temperature, but not by concentration or pressure.
What is a common mistake when calculating equilibrium constant?
A common mistake is to confuse the reaction quotient (Q) with the equilibrium constant (K).
How to avoid errors in calculating Kp and Kc?
Ensure that the units are consistent and that Δn is calculated correctly.
What is the relationship between equilibrium constant and Gibbs free energy?
The equilibrium constant is related to the Gibbs free energy change by ΔG = -RT ln(K).
How to derive the equilibrium constant expression?
The equilibrium constant expression can be derived from the law of mass action and the definition of equilibrium.










