Transport number for IIT JAM is a fundamental concept used to describe the movement of particles in a solution, critical for competitive exam students to grasp the underlying principles of transport phenomena.
Syllabus: Transport Number in IIT JAM Syllabus – Thermodynamics and Transport Phenomena
The topic of transport number is part of the Thermodynamics and Transport Phenomena unit in the IIT JAM syllabus, which is also relevant for CSIR NET and GATE exams.
Understanding the transference number (just another name for transport number) is essential for this unit. It is defined as the ratio of the rate of transport of a specific ion to the total rate of transport of all ions in an electrolytic solution.
Key textbooks that cover this topic include:
Transport Phenomena by Bird, Stewart, and Lightfoot
Physical Chemistry by Peter Atkins and Julio de Paula
The transport number is a critical concept in understanding various transport phenomena, such as diffusion, migration, and convection. Students preparing for IIT JAM, CSIR NET, and GATE exams should focus on grasping this concept and its applications.
What is Transport Number For IIT JAM: Understanding the Concept
Imagine you and a friend are moving boxes out of a warehouse. You are fast and carry 6 boxes every minute, while your friend moves 4 boxes a minute. Together, you are moving 10 boxes a minute. If someone asks, “What fraction of the work did you do?”, you’d say 6 out of 10, or 60%.
That is exactly what the transport number is, but for ions in a solution. When you apply an electric field, ions start racing toward opposite electrodes. The transport number is just the measure of an ion’s relative velocity. It tells us the ratio of the velocity of a specific ion to the total velocity of all the ions hanging out in that solution.
Because it is a share of the total work, the transport number is a dimensionless quantity that always sits between 0 and 1. The total sum of the transport numbers for all ions in the solution always adds up to exactly 1. If an ion has a transport number of 0.6, it means it is doing 60% of the heavy lifting to carry the current.
Two major factors change how fast these ions can run:
Concentration: How crowded the solution is.
Temperature: Heating things up gives the ions more kinetic energy, making them move faster and altering their mobility.
At VedPrep, we always remind students that mastering these fundamental shifts in ionic behavior is what helps you clear the tricky conceptual questions in the exam room.
Transport Number For IIT JAM: Worked Example
To link the transport number of an ion to its actual mobility, we can look at a straightforward ratio. For a 1:1 electrolyte like NaCl, you can find the transport number of Na+ ions (t+) using this formula:
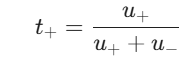
In this formula, u+ is the ionic mobility of the Na+ ions, and u– is the ionic mobility of the Cl– ions.
Let’s plug in the numbers. At infinite dilution, the ionic mobilities of Na+ and Cl⁻ ions are 5.19 × 10⁻⁸ m s⁻¹ V⁻¹ and 7.62 × 10⁻⁸ m2 s⁻¹ V⁻¹..
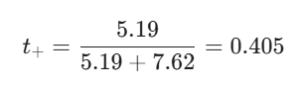
The transport number of Na⁺ ions is roughly 0.405. This tells us that Na⁺ ions carry about 40.5% of the total current through the solution, while the speedier Cl⁻ ions take care of the remaining 59.5%.
Common Misconceptions About Transport Number For IIT JAM
A massive trap that many IIT JAM aspirants fall into is assuming that the transport number is directly equal to the charge of the ion. It is easy to see a +2 or -1 charge and think that dictates everything, but that is a mistake. The transport number depends entirely on the fraction of the total current the ion carries, which is down to its speed, not its charge.
Think of it like a crowded market. A small, nimble person (like a small, highly hydrated ion) might weave through the crowd much slower than a larger, unhindered person, regardless of how much energy they started with.
Another frequent mix-up is treating transport number and ionic conductivity as the exact same thing. Here is the difference:
Ionic conductivity is the actual capacity of an ion to conduct electricity.
Transport number is just a fraction—the relative share an ion contributes to the total pool of current.
Keeping these terms separated in your mind will save you from silly mistakes on exam day.
Real-World Applications of Transport Number For IIT JAM
The concept of the transport number isn’t just something confined to your physical chemistry textbooks; it actually runs a lot of everyday industrial tech.
Take electrolysis as an example. When industries want to break down chemical compounds, knowing the transport number helps them figure out the overall efficiency of the cell. If they are tracking how fast hydrogen gas evolves, the transport number of the hydrogen ions gives them the exact rate of the reaction.
It is also a big deal in electroplating—the process used to coat jewelry or protect car parts with a thin layer of metal. To get a perfectly smooth, uniform coat of metal without any patchy spots, engineers have to balance the electrolyte mix. They use the transport number to make sure the right ions arrive at the surface at the perfect speed. Even in industrial chemical synthesis, understanding how these ions move helps engineers design reactors that don’t waste energy.
Key Formulas and Equations for Transport Number For IIT JAM
To secure those scoring marks in the physical chemistry section, you will want to be comfortable with a couple of core equations. First up is the Nernst-Einstein equation, which links the diffusion coefficient (D) of an ion to its charge (z), the gas constant (R), absolute temperature (T), and the Faraday constant (F):
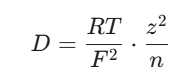
When you want to look at how this relates directly to how the current is shared, the transport number (t) can be framed alongside the diffusion coefficient like this:
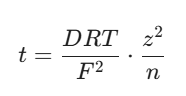
In these equations, n represents the total number of ions taking part in the transport process. Getting comfortable with twisting and turning these formulas during your study sessions will make numerical questions feel like second nature.
Additional Resources
This entire topic sits in the core physical chemistry section of the official IIT JAM syllabus under Electrochemistry. If you want to turn pages and dive into the deep theory, standard reference books like Atkins’ Physical Chemistry are excellent companions. For an even closer look at the mechanics of moving particles, Transport Phenomena by Bird, Stewart, and Lightfoot is a great read.
If you ever feel stuck on a tough derivation or want to see these concepts broken down visually, we have put together step-by-step video tutorials and exam-style mock tests over at VedPrep to help you test your speed and accuracy.
Final Thoughts
The concept of the transport number serves as a vital bridge between theoretical physical chemistry and its practical electrochemical applications. For IIT JAM 2027 aspirants, moving beyond simple formula memorization to understand the nuances of ionic mobility and environmental factors like temperature and concentration is key to scoring high in the Thermodynamics and Transport Phenomena section.
As you integrate these principles into your study routine—leveraging both classic textbooks and modern digital resources—you will find that a clear grasp of how ions “carry their weight” in a solution clarifies not just exam questions, but the very nature of chemical dynamics.
To know more in detail from our expert, watch our YouTube video:
Frequently Asked Questions
How is transport number related to ionic mobility?
The transport number is directly related to ionic mobility, as it depends on the mobility of the ions in the solution. Ions with higher mobility carry more current, resulting in a higher transport number.
What is the significance of transport numbers in electrochemistry?
The transport number is significant in electrochemistry as it helps in understanding the behavior of ions in solution, their interaction with the electrode, and the overall electrochemical reaction.
How is the transport number measured?
The transport number can be measured using the Hittorf method, which involves measuring the change in concentration of ions at the electrodes during electrolysis.
What are the limitations of the Hittorf method?
The Hittorf method has limitations, such as assuming that the ions do not interact with each other or with the solvent, and that the electrode reactions are reversible.
What is the relationship between transport number and ionic conductivity?
The transport number is related to ionic conductivity, as it determines the contribution of each ion to the overall conductivity of the solution.
How does temperature affect transport numbers?
Temperature affects the transport number by changing the mobility of ions, with higher temperatures generally increasing ionic mobility and transport numbers.
How is transport number relevant to IIT JAM Physical Chemistry?
Transport number is a crucial concept in Physical Chemistry, and is frequently asked in IIT JAM. Understanding transport numbers and their applications is essential for solving problems in electrochemistry.
What types of problems are asked in IIT JAM regarding transport numbers?
In IIT JAM, problems related to transport number may involve calculating transport numbers, understanding their relationship with ionic mobility, and applying them to electrochemical reactions.
How can I practice transport number problems for IIT JAM?
Practice problems from previous years' papers, and use online resources such as VedPrep to get familiar with different types of questions and to improve problem-solving skills.
What are common mistakes made when calculating transport numbers?
Common mistakes include incorrect assumptions about ionic interactions, neglecting to account for electrode reactions, and using incorrect values for ionic mobility.
How can I avoid mistakes when applying transport numbers to electrochemical reactions?
Carefully consider all factors affecting transport numbers, such as temperature and ionic interactions, and ensure that the correct equations and values are used.
What is the relationship between transport number and activity coefficient?
The transport number is related to the activity coefficient, as it affects the thermodynamic activity of ions in solution, which in turn influences transport numbers.
How does transport number relate to non-equilibrium thermodynamics?
Transport numbers play a crucial role in non-equilibrium thermodynamics, as they help describe the behavior of ions in systems far from equilibrium.
What are recent developments in the study of transport numbers?
Recent developments include studies on transport numbers in complex systems, such as ionic liquids and biological systems, and the development of new experimental techniques to measure transport numbers.










