Transition state theory is a crucial concept in physical chemistry that explains the rate of chemical reactions by considering the energy barrier and the probability of forming a transition state. Understanding this theory is essential for students appearing for IIT JAM to tackle complex reaction kinetics questions.
Syllabus: Physical Chemistry Unit – Reaction Kinetics and Mechanism
In the official IIT JAM syllabus, this topic sits firmly under chemical kinetics. If you want to master this unit, your study checklist should look like this:
Reaction rates and rate constants: Knowing how fast a reaction goes and what factors tweak that speed.
Activation energy and its significance: The molecular price of admission for a reaction.
Collision theory and its limitations: Why just smashing molecules together isn’t enough to explain complex reaction speeds.
When it comes to picking your study material, classic textbooks like Atkins’ Physical Chemistry are excellent for building a solid theoretical foundation. If you want to push your problem-solving limits, flipping through Irodov’s Problems in General Physics can actually give you a unique edge with mathematical modeling. Over at VedPrep, we also map out these exact tricky areas to help you connect textbook theory with actual exam-style questions.
Transition State Theory: A Comprehensive Overview
Let’s break down Transition State Theory without the heavy academic jargon. Think of a chemical reaction as a hike over a steep mountain. The reactants are at the base camp on one side, and the products are the destination on the other side.
To get to the other side, the hikers have to climb all the way to the absolute highest point of the pass. In chemistry, that highest point is what we call the transition state or the saddle point. It is a fleeting, highly unstable arrangement of atoms where old bonds are halfway broken and new bonds are halfway formed.
Here are the core concepts you need to keep straight:
Transition state: A temporary, high-energy state where reactants are in limbo, deciding whether to turn into products or fall back to where they started.
Activation energy (Ea): The exact amount of energy difference between your starting reactants and that peak.
Energy barrier: The minimum energetic hill a reaction must climb to actually happen.
At VedPrep, we like to remind students that a lower energy barrier means more molecules can clear the hurdle easily, leading to a faster reaction. If the hill is too high, the reaction slows to a crawl.
Transition state theory For IIT JAM: Key Concepts and Formulas
When you face IIT JAM questions, you will need to bridge the gap between thermodynamics and kinetics. Let’s look at the math that binds them together.
First, remember Gibbs free energy change (ΔG) from your thermodynamics classes. It tells us if a reaction wants to happen spontaneously. The equation looks like this:
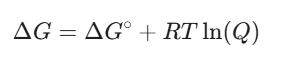
Where ΔG° is the standard Gibbs free energy change, R is the gas constant, T is the temperature, and Q is your reaction quotient.
But TST takes this further by introducing the Gibbs free energy of activation (![]() ), which is the energy needed to reach the transition state itself. To find out how fast a reaction goes, we look at the Arrhenius equation, which connects the rate constant (k) to the activation energy (Ea):
), which is the energy needed to reach the transition state itself. To find out how fast a reaction goes, we look at the Arrhenius equation, which connects the rate constant (k) to the activation energy (Ea):
![]()
In this formula, A is the pre-exponential factor. TST explains that k is directly tied to the stability of that transition state. If the transition state is highly ordered or difficult to form, the reaction rate drops.
| Key Concept | Description |
| Gibbs free energy | The energy change that tells us if a reaction can happen under constant temperature and pressure. |
| Transition state | The unstable, peak-energy setup where old bonds stretch and new ones start to blink into existence. |
| Activation energy | The minimum energetic punch required to get reactants over the hill. |
Worked Example: Applying Transition State Theory to a Chemical Reaction
Let’s see how this plays out in a typical problem. Imagine a fictional reaction where two molecules, let’s call them A and B, need to collide to form products C and D:
Let’s say the activation energy (Ea) for this reaction is $100\text{ kJ/mol}$ (which is 100,000 J/mol). We are running this at a room temperature of $300\text{ K}$, and the gas constant R is 8.314 J/(mol· K). Assuming our pre-exponential factor A is 106 M-1s-1, what is the rate constant $k$?
Let’s plug the numbers into our Arrhenius equation:
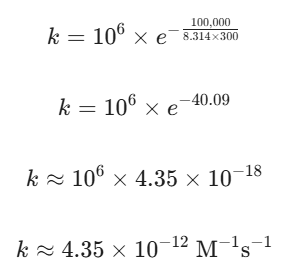
Because that energy barrier is quite high (100 kJ/mol) relative to room temperature, only a tiny fraction of molecular collisions have enough energy to reach the transition state. This calculation shows exactly why reaction rates are incredibly sensitive to both temperature changes and the height of the activation energy barrier.
Additional Resources for Learning Transition State Theory
Getting a true handle on TST means going beyond basic calculations. You want to get comfortable with the Eyring equation, which uses statistical mechanics to predict reaction rates based on the entropy and enthalpy of the transition state.
To get your prep on track, we suggest a balanced mix of resources:
Standard Textbooks: Treat Physical Chemistry by Atkins and Friedman as your conceptual anchor.
VedPrep Study Material: We provide targeted video lectures, specialized question banks, and crisp notes designed around real exam trends to save you time.
Peer Groups & Forums: Joining online study communities, like the VedPrep community forums, lets you chat about tricky doubts with other students who are fighting the same battle.
Taking a look at free online lectures or solved question breakdowns can quickly clear up misconceptions before they stick.
Conclusion
Mastering Transition State Theory is a massive step toward nailing the chemical kinetics portion of your IIT JAM exam. By connecting the dots between thermodynamic stability and actual reaction speeds, TST gives you the exact tools you need to break down complex molecular transformations.
As you keep pushing forward with your prep, focus on getting highly comfortable with energy profile diagrams and the math behind the Eyring equation. Keep practicing, make the most of the tools and practice sets we offer at VedPrep , and you will turn this challenging topic into a major score booster on exam day.
Frequently Asked Questions
What is the significance of the transition state?
The transition state is crucial as it determines the energy required for a reaction to occur. It's a temporary state where reactants are in the process of transforming into products, and its energy is the activation energy.
How does TST relate to chemical kinetics?
TST provides a framework for understanding the rates of chemical reactions. It helps in determining the rate constant and activation energy, which are essential in chemical kinetics.
What are the assumptions of Transition State Theory?
TST assumes that the transition state is in equilibrium with the reactants and that the partition function of the transition state can be separated into partition functions of the reactants and a vibrational partition function.
What is the role of the partition function in TST?
The partition function is used to calculate the thermodynamic properties of the transition state. It's a measure of the number of available states in the system, and it plays a crucial role in determining the rate constant.
How does TST explain the effect of temperature on reaction rates?
TST explains that an increase in temperature provides more energy for the reactants to overcome the activation energy barrier, thus increasing the reaction rate.
What are the limitations of Transition State Theory?
TST has limitations, such as assuming a single transition state and neglecting non-equilibrium effects. It also assumes that the reaction coordinate is a straight line, which may not always be the case.
What is the relationship between TST and the Arrhenius equation?
The Arrhenius equation is a phenomenological equation that describes the temperature dependence of reaction rates. TST provides a theoretical framework for understanding the Arrhenius equation and its parameters, such as the activation energy.
How does TST relate to the concept of catalysis?
TST explains how catalysts work by lowering the activation energy required for a reaction to occur. This allows the reaction to proceed faster and more efficiently.
What is the significance of TST in physical chemistry?
TST is a fundamental concept in physical chemistry that provides a deep understanding of chemical reactions and kinetics. It's widely used in various fields, including chemistry, materials science, and biology.
How can I apply TST to solve problems in IIT JAM?
To apply TST in IIT JAM, focus on understanding the concepts and formulas. Practice problems related to calculating rate constants, activation energy, and reaction rates using TST.
What types of questions can I expect in IIT JAM related to TST?
Expect questions on the fundamental concepts of TST, such as the definition of transition state, activation energy, and partition function. Also, be prepared for numerical problems involving the application of TST formulas.
What are common mistakes students make when applying TST?
Common mistakes include incorrect calculation of partition functions, misunderstanding the concept of transition state, and neglecting the assumptions of TST.
What are some recent developments and applications of TST?
Recent developments include the application of TST to complex systems, such as enzymatic reactions and materials science. TST is also used in combination with other techniques, such as quantum mechanics and molecular dynamics.
How can TST be used in combination with other theories?
TST can be combined with other theories, such as quantum mechanics and statistical mechanics, to provide a more comprehensive understanding of chemical reactions. This helps in understanding complex systems and phenomena.










