This piece explores the core ideas of atomic makeup, molecule creation, and the ways elements connect, offering complete knowledge vital for those preparing for the CSIR NET exam, specifically addressing Structure of atoms, molecules and chemical bonds For CSIR NET.
Structure of atoms, molecules and chemical bonds For CSIR NET
The subject matter regarding the makeup of atoms, molecules, and chemical linkages falls under Unit 1 of the CSIR NET syllabus, as formally documented by the National Testing Agency (NTA). This section is essential for grasping the groundwork of chemistry, particularly relevant to the CSIR NET exam. The outlined content encompasses vital domains critical for CSIR NET study.
For detailed exploration, learners might consult established texts like Physical Chemistry by P.W. Atkins and J. de Paula. This volume thoroughly addresses subjects such as the physical state, molecular arrangement, and chemical linkage, all vital for CSIR NET readiness, especially in grasping atomic operations, compounds, and chemical unions pertinent to CSIR NET. These areas lay a firm groundwork for more complex chemical principles.
The key topics under this section include:
- Atomic makeup: Grasping atom constituents, such as electrons, protons, and neutrons, for CSIR NET.
- Molecular framework: Examining atom placement inside molecules, molecules, and chemical linkages for CSIR NET.
- Bonding chemistry: Investigating the attractions that unite atoms within molecules, encompassing covalent, ionic, and metallic connections, molecules, and chemical bonds for CSIR NET.
Grasping this area thoroughly is key for excelling on the test. This material forms the bedrock for higher-level chemistry ideas, notably molecule structures and chemical linkages within this specific domain, particularly for the CSIR NET exam. A solid comprehension of these principles aids in deciphering numerous chemical occurrences.
Atomic Structure
The makeup of an atom involves three main parts: protons, neutrons, and electrons, which are essential for this subject regarding CSIR NET. Protons and neutrons are located in the core, whereas electrons circle this center. As per molecules and chemical bonds For CSIR NET, the count of protons within an atom’s nucleus sets the element’s nature, a vital notion in the atomic structure, molecules, and chemical bonds for CSIR NET. Grasping this idea is paramount for comprehending chemical characteristics.
The ordering of electrons, termed electron configuration, is vital for grasping this area, encompassing molecules and chemical bonds relevant to CSIR NET. This structure is directed by a pair of core tenets: the Aufbau principle and the Pauli exclusion principle. The Aufbau principle dictates that electrons fill the lowest energy states first. Conversely, the Pauli exclusion principle maintains that no more than two electrons, possessing opposing spins, can occupy any single orbital, both factors being significant for this field, molecules, and chemical bonds in the context of CSIR NET.
Molecular Structure and Chemical Bonding
A molecule’s make-up is key to figuring out its traits and how it acts, especially concerning molecules and chemical bonds relevant to CSIR NET. Electron dot diagrams, or Lewis structures, illustrate how electrons are positioned within a molecule, aiding in forecasting molecular shape and charge distribution within the study of Structure of atoms, molecules and chemical bonds for CSIR NET. Grasping this is essential for chemistry.
VSEPR (Valence Shell Electron Pair Repulsion) theory helps forecast molecular shapes by considering the repulsion among electron groupings, molecules, and chemical linkages, particularly relevant for CSIR NET. This principle holds that these electron groupings surrounding a core atom will position themselves to lessen repulsion, consequently yielding a defined molecular structure, molecules, and chemical bonds For CSIR NET.
Intermolecular Forces and Polarity
Attractive and repulsive forces existing between molecules are termed as intermolecular forces, and they significantly impact the behavior of atoms. Various kinds of these forces exist, such as London dispersion forces (also referred to as van der Waals forces) and hydrogen bonds, all pertinent to the CSIR NET examination. These interactions are fundamental in establishing the physical characteristics of materials in Structure of atoms, molecules and chemical bonds.
Worked Example: Bond Order and Molecular Orbital Theory
Molecular Orbital (MO) theory is a potent method for grasping the electronic makeup of chemical species and the nature of their linkages, relevant for CSIR NET. A central idea within MO theory is bond order, which quantifies the extent of electron participation in the connections between atoms within a compound, directly tied to the Structure of atoms, molecules and chemical bonds for CSIR NET. The equation for calculating bond order is: Bond Order = (count of bonding electrons – count of antibonding electrons) / 2, an expression vital for comprehending atomic architecture.
Misconception: Common Mistakes in Chemical Bonding
Learners frequently suppose all chemical linkages are ionic, which is an incorrect notion within this subject area. Chemical connections broadly fall into three categories: ionic, covalent, and metallic, regarding molecules and chemical bonds for CSIR NET. Grasping these distinctions is vital for a solid footing in chemistry.
Application: Real-World Examples
The formation of chemical bonds is fundamental to grasping the characteristics and actions of substances, directly tied to how atoms operate for CSIR NET. Sharing electrons among atoms is evident in covalent bonds, such as those found in water (H2O) and diamond (pure carbon), key examples in the realm of atoms, molecules, and chemical bonds for CSIR NET. Conversely, the movement of electrons from one atom to another defines ionic bonds, exemplified by sodium chloride (NaCl), which highlights another vital area concerning Structure of atoms, molecules and chemical bonds.
Key Concepts and Formulas
The topic of molecules and chemical bonds is foundational to chemistry, essential for grasping diverse chemical occurrences, particularly in the context of CSIR NET preparation. Coulomb’s law outlines the electrostatic force between charged entities, for instance, electrons and atomic nuclei. The corresponding equation is:
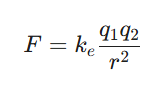
Practice and Preparation for CSIR NET
To excel in the subject of atomic structure for CSIR NET, a comprehensive grasp of the foundational concepts, molecules, and chemical bonds is necessary for CSIR NET. A solid command of atomic architecture, molecular shapes, and the nature of chemical bonds is vital for achieving success in CSIR NET. Working through practice exercises and previous examination papers can aid in solidifying these ideas.
Conclusion
Understanding the arrangement of atoms, how molecules form, and the nature of chemical bonds, as covered in the CSIR NET syllabus, is absolutely essential for exam candidates. As per Structure of atoms, molecules and chemical bonds, this requires a thorough grasp of the underlying theories and practical uses. Through diligent study of atomic composition, molecular frameworks, and the forces holding atoms together—all tailored to the CSIR NET curriculum on atomic structure, molecules, and chemical bonds—learners can significantly boost their expertise and prospects for passing the test.
Conquering this subject for CSIR NET goes beyond simple rote learning of electron setups or shape predictions; it involves cultivating a feel for space regarding how the tiny realm governs large-scale chemical actions. As we’ve seen, a thorough look at quantum descriptors, molecular orbital concepts, and forces between molecules offers the necessary critical structure to handle the intricate, layered problems usual in the Unit 1 material. Excelling in the 2026 test necessitates moving from fundamental grasp to sophisticated use, enabling accurate forecasting of molecular unevenness and linkage strengths.
To enhance your readiness further and gain specialized instruction focused on these distinct chemical details, leveraging materials from VedPrep can offer the advantage required to thrive. By regularly applying these core concepts, you secure a solid groundwork that will bolster your performance throughout the Physical and Inorganic chemistry sections.
To know more in detail from our expert faculty, watch our YouTube video:
Frequently Asked Questions (FAQs)
What is the basic structure of an atom?
An atom consists of a nucleus containing protons and neutrons, surrounded by electrons in orbitals. The nucleus has a positive charge, while electrons have a negative charge, resulting in a neutral atom.
What are molecules and how are they formed?
Molecules are groups of two or more atoms chemically bonded together. They are formed when atoms share or exchange electrons to form chemical bonds, resulting in a stable molecule with a specific shape and properties.
What are the different types of chemical bonds?
There are several types of chemical bonds, including ionic bonds, covalent bonds, metallic bonds, and hydrogen bonds. Each type of bond has distinct characteristics and plays a crucial role in determining the properties of molecules.
What is the significance of molecular structure in biology?
The structure of molecules is crucial in biology as it determines their function and interaction with other molecules. The shape and properties of molecules influence their role in biological processes, such as enzyme-substrate interactions and protein folding.
How do atoms and molecules interact with each other?
Atoms and molecules interact through chemical bonds, which are formed when atoms share or exchange electrons. These interactions determine the properties of molecules and their behavior in different biological and chemical contexts.
What is the role of chemical bonds in molecular stability?
Chemical bonds play a crucial role in determining the stability of molecules. The type and strength of chemical bonds influence the energy required to break or form bonds, which in turn affects the stability and reactivity of molecules.
How do molecular interactions relate to biological processes?
Molecular interactions are essential for various biological processes, such as enzyme-substrate interactions, protein-protein interactions, and cell signaling. These interactions determine the behavior and function of molecules in living organisms.
What is the relationship between molecular structure and function?
The structure of a molecule determines its function and interaction with other molecules. Understanding the relationship between molecular structure and function is crucial for predicting and manipulating molecular behavior in various biological and chemical contexts.
What are the key factors that determine molecular stability?
The key factors that determine molecular stability include the type and strength of chemical bonds, molecular geometry, and electronic properties. Understanding these factors is crucial for predicting and manipulating molecular behavior in various biological and chemical contexts.
How can I apply knowledge of atomic structure to CSIR NET questions?
Understanding atomic structure is essential for answering questions related to chemical bonding, molecular interactions, and biological processes. Focus on applying concepts to solve problems and analyze data in the context of CSIR NET syllabus.
How can I use knowledge of atomic and molecular structure to solve problems in CSIR NET?
To solve problems in CSIR NET, apply knowledge of atomic and molecular structure to analyze data and predict molecular behavior. Focus on understanding the underlying concepts and mechanisms that govern molecular interactions and biological processes.
How can I avoid mistakes in identifying chemical bonds?
To avoid mistakes in identifying chemical bonds, carefully analyze the molecular structure and electron configuration. Understand the characteristics of different types of bonds and practice identifying them in various molecules.
What are some advanced topics in atomic and molecular structure?
Advanced topics in atomic and molecular structure include quantum mechanics, molecular orbital theory, and computational chemistry. These topics provide a deeper understanding of atomic and molecular behavior and are relevant to cutting-edge research.
How can I apply knowledge of molecular structure to complex biological systems?
To apply knowledge of molecular structure to complex biological systems, focus on understanding the interactions between multiple molecules and their role in biological processes. Use computational tools and theoretical frameworks to analyze and predict molecular behavior.
What are some recent developments in understanding chemical bonds and molecular interactions?
Recent developments in understanding chemical bonds and molecular interactions include advances in computational chemistry, single-molecule spectroscopy, and structural biology. These advances have improved our understanding of molecular behavior and its role in biological processes.














