Metalloenzymes are specialized proteins containing a tightly bound metal ion cofactor essential for catalytic activity. These biological catalysts facilitate critical life processes such as respiration, photosynthesis, and nitrogen fixation by stabilizing transition states and enabling redox reactions. In Bioinorganic Chemistry, Metalloenzymes represent the functional integration of metallic elements into organic protein frameworks to achieve high reaction specificity.
Defining Metalloenzymes within Bioinorganic Chemistry
Metalloenzymes function as biological catalysts where the metal ion serves as an integral part of the protein structure. Unlike metal activated enzymes which have loosely bound ions, Metalloenzymes retain their metal components even after purification processes. The metal ion usually resides in a coordination pocket where it interacts with amino acid side chains like histidine, cysteine, or glutamate.
This interaction modifies the chemical properties of both the metal and the protein to enable complex transformations. You will find that approximately one third of all known enzymes require metal ions for their function. This field is a core pillar of Bioinorganic Chemistry and remains a vital topic for students preparing for the RPSC Assistant Professor Chemistry Paper II. Understanding the distinction between structural and catalytic metal sites is fundamental for mastering this subject.
The Structural Classification of Metalloenzymes
The classification of Metalloenzymes depends on the specific metal ion present and its coordination geometry. Common metals include iron, copper, zinc, magnesium, and manganese. Iron based systems like Cytochrome P450 facilitate oxidation, while Zinc based systems like Carbonic Anhydrase manage hydration reactions. Each metal brings unique redox potentials and Lewis acid strengths to the active site.
Zinc is a preferred Lewis acid in Metalloenzymes because it lacks redox activity under biological conditions, preventing unwanted side reactions. These structural nuances are frequently tested in the RPSC Assistant Professor Chemistry Paper II. You should note that the protein environment exerts an entatic state, which is a pre distorted geometry that lowers the activation energy for catalysis. This concept explains why biological catalysts are often more efficient than synthetic laboratory analogs.
The Kinetic Power of Carbonic Anhydrase
Carbonic Anhydrase is a quintessential example of a Zinc Metalloenzyme that manages the interconversion of carbon dioxide and bicarbonate. The Zinc ion is bound by three histidine side chains and a water molecule or hydroxide anion. This structure promotes the swift nucleophilic strike on carbon dioxide.
This enzyme’s turnover figure hovers around one million reactions each second. Such great efficacy is essential for sustaining the pH equilibrium and CO2 conveyance within human blood. In Bioinorganic Chemistry, this enzyme demonstrates how a metal ion can lower the pKa of bound water from 14 to about 7. This modification enables the creation of a dense amount of hydroxide anion at body-level pH values. Those studying for the RPSC Assistant Professor Chemistry Paper II ought to commit the specific configuration of this complex and the precise function of the Zinc(II) ion to memory.
Numerical Expressions and Data in Enzyme Kinetics
Assessing the effectiveness of Metalloenzymes necessitates familiarity with particular numerical values and formulas. The link between substrate amounts and the pace of reaction is described by the Michaelis-Menten kinetic model. For numerous Metalloenzymes, catalytic effectiveness is conveyed by dividing the turnover number by the Michaelis constant. This is shown as:
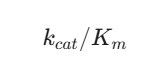
Another important numerical expression involves the calculation of the reduction potential of the metal center using the Nernst Equation. For a redox active metalloenzyme, the potential E is calculated as:
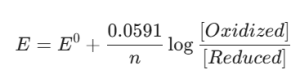
These figures suggest the thermodynamic possibility of electron movement in metalloproteins such as Ferredoxins or Cytochromes. For the RPSC Assistant Professor Chemistry Paper II, understanding how to interpret these specific metrics is crucial for tackling questions concerning Bioinorganic Chemistry and how enzymes function.
Key Theories and Principles in Metalloenzyme Function
A number of well-regarded frameworks account for the exceptional catalytic skill of Metalloenzymes . These concepts link the fields of inorganic chemistry and molecular biology. The main theoretical models used for these systems are outlined in the table below.
| Theory/Principle | Core Concept | Application in Metalloenzymes |
|---|---|---|
| Entatic State Theory | Pre-distorted coordination geometry at the active site. | Lowers activation energy for electron transfer in Blue Copper Proteins. |
| Lewis Acid Catalysis | Metal ion polarizes a substrate or stabilizes a negative charge. | Zinc(II) in Carboxypeptidase A and Carbonic Anhydrase. |
| Redox Potential Tuning | Protein ligands shift the reduction potential of the metal ion. | Iron-Sulfur clusters in the electron transport chain. |
| Hard-Soft Acid-Base (HSAB) | Preference of metals for specific donor atoms (O, N, S). | Predicting metal binding to amino acid side chains. |
The Role of Iron in Oxygen Transport and Storage
Iron is perhaps the most versatile metal in Bioinorganic Chemistry, appearing in Hemoglobin, Myoglobin, and various Cytochromes. In Hemoglobin, the iron is in the +2 oxidation state and is coordinated to a porphyrin ring and a proximal histidine. When oxygen binds, the iron moves into the plane of the porphyrin, triggering a conformational change in the protein.
This cooperative binding is a hallmark of Metalloenzymes that function as transporters rather than pure catalysts. You must distinguish between heme iron and non heme iron proteins. Non heme iron enzymes, such as Hemerythrin, use different coordination strategies to achieve similar oxygen binding goals. Proficiency in these iron based systems is a major requirement for the RPSC Assistant Professor Chemistry Paper II, as they represent the intersection of coordination geometry and physiological function.
Contrarian Perspectives on Metal Substitution
A frequent belief in Bioinorganic Chemistry suggests that the precise metal inherent to a native metalloenzyme is the sole one capable of enabling its role. Nevertheless, findings from laboratory studies frequently demonstrate that swapping the native metal for an alternative can, at times, preserve or even boost performance. For instance, substituting Zinc with Cobalt in specific enzymes offers a spectroscopic tool while not entirely eliminating catalytic capability.
A drawback of this method is that controlled lab settings don’t mirror the intricate metal balance within a living cell. Even if an enzyme with Cadmium substitution functions okay in a beaker, the cell would perceive Cadmium as harmful. Consequently, the selection of metals in nature is dictated by their accessibility and evolutionary forces, not solely by chemical responsiveness. This perspective highlights the difference between chemical possibility and biological reality.
Practical Application in Environmental Detoxification
Metalloenzymes are directly involved in environmental science via the method of bioremediation. Specific bacteria generate Metalloenzymes that have the capacity to degrade harmful organophosphates or convert heavy metal ions into safer states. As an illustration, the enzyme organophosphate hydrolase utilizes a pair of Zinc or Manganese centers to neutralize pesticides.
Grasping these processes enables researchers to formulate biomimetic catalysts for industrial waste management. This hands-on utilization of Bioinorganic Chemistry demonstrates that Metalloenzymes are more than just abstract concepts; they are instruments for tackling contemporary ecological issues. Those taking the RPSC Assistant Professor Chemistry Paper II utilize these illustrations to highlight the practical significance of coordination chemistry.
Strategic Importance for Competitive Exams
For candidates aiming for the RPSC Assistant Professor Chemistry Paper II, Metalloenzymes are a high yield topic. You must focus on the active site structure, the oxidation state of the metal, and the specific ligands involved. VedPrep helps students master these details to qualify for the RPSC Assistant Professor exam.
The organization consistently has a history of achieving top national rankings, including the highest score each year, through offering profound comprehension of Bioinorganic Chemistry. Excelling in this field necessitates a blend of recalling biological functions and grasping the fundamental inorganic concepts. Concentrating on the electron configuration of the metallic core will offer the necessary illumination to successfully tackle intricate objective questions.
Conclusion
Grasping the chemical tenets of Metalloenzymes is a crucial hurdle for applicants aspiring to excel in the RPSC Assistant Professor Chemistry Paper II. These biological accelerators showcase the intricate melding of metallic elements inside protein frameworks to execute essential life processes with superb swiftness. By concentrating on the bond arrangements, electrical potentials, and speed-related concepts detailed in this material, you can establish the solid theoretical base necessary for advanced scholastic assessments. VedPrep continues to commit to assisting your path to an academic post through expert viewpoints and thorough learning aids. Ongoing study of these inorganic-biological frameworks will both bolster your test readiness and broaden your respect for nature’s molecular workings.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions (FAQs)
How do Metalloenzymes differ from metal-activated enzymes?
The primary difference lies in the strength of the metal-protein bond. Metalloenzymes hold their metal ions tightly, often retaining them through purification. Metal-activated enzymes bind ions loosely from the surrounding solution. In Metalloenzymes, the metal is a permanent structural fixture, whereas in metal-activated systems, the ion often acts as a transient modulator.
Which metal ions are most common in Metalloenzymes?
Transition metals dominate this field due to their variable oxidation states and coordination geometries. Iron, Zinc, Copper, Manganese, and Magnesium are the most prevalent. Zinc is frequently used for non-redox catalysis, while Iron and Copper are favored for electron transfer and oxygen transport due to their ability to cycle between oxidation states.
What is the role of Zinc in Carbonic Anhydrase?
In Carbonic Anhydrase, Zinc(II) acts as a powerful Lewis acid. It coordinates with a water molecule, significantly lowering its pKa from 14 to approximately 7. This creates a high local concentration of hydroxide ions at physiological pH, enabling the rapid conversion of carbon dioxide into bicarbonate.
How does the Nernst Equation apply to Metalloenzyme study?
The Nernst Equation allows researchers to calculate the reduction potential of a redox-active metal center within a protein. Since the surrounding amino acid residues can shift the metal's electron affinity, this calculation is vital for understanding how enzymes like Cytochromes or Ferredoxins participate in the electron transport chain.
How do ligands influence Metalloenzyme activity?
Ligands, such as histidine, cysteine, and glutamate side chains, coordinate directly with the metal ion. They determine the metal's electronic environment, redox potential, and available coordination sites. A change in a single ligand can completely alter the enzyme's substrate specificity or its ability to undergo redox cycling.
What is the significance of the Michaelis-Menten constant for these enzymes?
The Michaelis-Menten constant (Km) indicates the affinity of a Metalloenzyme for its substrate. A lower Km suggests high affinity, meaning the enzyme reaches maximum catalytic rate at lower substrate concentrations. This numerical value is a key metric for evaluating the efficiency of biological catalysts in RPSC-level chemistry problems.
How is oxygen transport coordinated in Hemoglobin?
Hemoglobin utilizes an Iron(II) protoporphyrin IX unit. When oxygen binds to the sixth coordination site of the iron, it triggers a shift of the iron atom into the porphyrin plane. This mechanical movement is transmitted through the protein chain, increasing the oxygen affinity of neighboring subunits through cooperative binding.
Why does metal substitution sometimes fail to maintain enzyme activity?
While substituting one metal for another (e.g., Cobalt for Zinc) can provide spectroscopic data, it often reduces catalytic efficiency. The protein fold is evolved to match the specific ionic radius and preferred geometry of the native metal. A mismatch can disrupt the active site's delicate electronic balance, rendering the enzyme inactive.
How does pH affect Metalloenzyme stability?
pH changes can protonate or deprotonate the amino acid ligands coordinating the metal ion. If a ligand like histidine becomes protonated, it may lose its ability to bind the metal, causing the ion to dissociate. This leads to the irreversible denaturation of the Metalloenzyme and loss of function.
What are the limitations of biomimetic models in Bioinorganic Chemistry?
Biomimetic models are small molecules designed to mimic the active site of a Metalloenzyme. While they provide insights into coordination chemistry, they lack the massive protein "scaffold" that provides the entatic state. Consequently, synthetic models rarely match the extreme speed and specificity of natural enzymes.
How do heavy metals cause Metalloenzyme inhibition?
Toxic heavy metals like Lead, Mercury, or Cadmium can displace native essential metals through competitive binding. Because these toxic ions have different sizes and electronic properties, they lock the enzyme in an inactive state. This molecular displacement is the primary mechanism behind many types of heavy metal poisoning.
What are the redox-tuning capabilities of Iron-Sulfur clusters?
Iron-Sulfur clusters can vary their reduction potentials over a wide range by changing their cluster geometry (e.g., [2Fe-2S] vs. [4Fe-4S]) and the nature of their protein environment. This allows the same basic metal unit to function at different steps of the electron transport chain.
How do Blue Copper Proteins achieve high-speed electron transfer?
Blue Copper Proteins, like Plastocyanin, utilize a highly distorted tetrahedral geometry for the Copper ion. This geometry is intermediate between the preferred states of Cu(I) and Cu(II). By minimizing the structural rearrangement needed during electron exchange, the enzyme achieves near-instantaneous redox transitions.
What is the evolutionary significance of Magnesium in kinases?
Magnesium is almost universally required for enzymes that handle ATP. It coordinates with the negatively charged phosphate groups, neutralizing their charge and positioning the phosphorus atom for nucleophilic attack. This fundamental interaction is essential for all energy-consuming biological processes.










