Peptide Bonds and Ramachandran Plots: Understanding the Structural Logic of Life in 2026 Structural biology has changed a lot since 2026.
AI tools like Alpha Fold-X and Rose TTA Fold are now so good at predicting protein structures that they can do it in seconds with almost atomic accuracy. But even with these big advances in technology, the basic rules of biology are still the same.
The “source code” of protein folding still depends on two important things: the strength of peptide bonds and the geometry that the Ramachandran Plot allows.
It is no longer enough for a student getting ready for CSIR NET Life Sciences, GATE, or IIT JAM to just memories definitions.
You need to know why and how. Why can’t a protein take on any shape? How do the electronic properties of peptide bonds determine the three-dimensional structure of enzymes and antibodies?
This long guide will take us from the quantum chemistry of the bond to the plot’s spatial mapping. Instead of just using the definitions from old textbooks, we’ll look at how these ideas are the foundation of modern synthetic biology and drug design.
The Basic Connection: How Peptide Bonds Work
Proteins do a lot of work in cells, but they are made up of a linear alphabet of amino acids. Peptide Bonds are what make this linear string into a working 3D machine. They are the magic that makes it work.
The Reaction of Condensation
The formation of peptide bonds is a dehydration synthesis (or condensation) reaction. It happens when the $\alpha$-carboxyl group ($-\text{COOH}$) of one amino acid meets the $\alpha$-amino group ($-\text{NH}_2$) of another.
The Process: A molecule of water ($H_2O$) is removed.
The outcome is a covalent bond ($-\text{CO}-\text{NH}-$).
This may seem simple, but in the biological context of 2026, we see this as an energy-dependent process done by the ribosome, which is the cell’s factory. Peptide bonds hold a lot of energy, so they are stable in terms of motion but not in terms of heat (meaning they won’t break on their own in your body, but they will when they do).
The Resonance Phenomenon: Why It Matters
This is the most important idea for tests. Peptide bonds are not just single bonds. They show “Resonance.”
The Nitrogen atom’s lone pair of electrons spreads out into the Carbonyl ($C=O$) group.
Partially Double Bond Character: This delocalization makes the $C-N$ bond about 40% double-bonded.
The Result: Double bonds can’t turn. So, the Peptide Bonds are flat and stiff. The six atoms that make up the molecule ($C_\alpha, C, O, N, H, C_\alpha$) are all in the same flat plane.
This flatness is what makes protein folding easier. Proteins would be floppy, shapeless chains if peptide bonds could rotate freely. Life as we know it would not exist.
Cis and Trans Configuration
The bond is stiff, so the groups that are attached to it can be frozen in certain places.
Trans Configuration: The two $\alpha$-carbons are on different sides of the Peptide Bonds. This is very likely (99.6% of bonds) because it reduces steric hindrance (clashing) between the side chains.
Cis Configuration: The α-carbons are on the same side. This makes things very crowded.
The Proline Exception: Proline is the bad guy. The energy difference between Cis and Trans is small because its side chain loops back to make a ring with its own nitrogen. About 10% of peptide bonds that involve proline are cis, which means they make “kinks” or turns in the protein chain.
The Geometry of Freedom: Angles of Torsion
Because peptide bonds are rigid plates, the protein chain can only bend at certain places between these plates. Think of a chain of stiff playing cards that are connected by metal rings. The rings (alpha-carbons) let things turn, but the cards (peptide units) don’t bend.
“Torsional Angles” or “Dihedral Angles” are terms that describe these points of rotation.
Phi ($\phi$) and Psi ($\psi$)
Each amino acid residue in a polypeptide chain has two bonds that can rotate around the central $\alpha$-carbon ($C_\alpha$):
Phi ($\phi$): The angle at which the Nitrogen ($N$) and the Alpha Carbon ($C_\alpha$) turn. This makes the amino group spin around the rest of the chain.
Psi ($\psi$): The angle of rotation between the Carbonyl Carbon ($C$) and the Alpha Carbon ($C_\alpha$). This makes the carboxyl group spin.
These angles could, in theory, be anywhere from $-180^\circ$ to $+180^\circ$. If this were the case, a protein could take on an infinite number of shapes. But in real life, the laws of physics get in the way.
G.N. Ramachandran, the Map Maker, comes in.
In the 1960s, Indian physicist G.N. Ramachandran asked a simple question: “What combinations of $\phi$ and $\psi$ are really possible?”
He thought of atoms as hard spheres that couldn’t be in the same place at the same time, like billiard balls. If you change the values of the $\phi$ and $\psi$ angles, the atoms in the side chains or the backbone will hit each other. This is known as steric hindrance or steric clash.
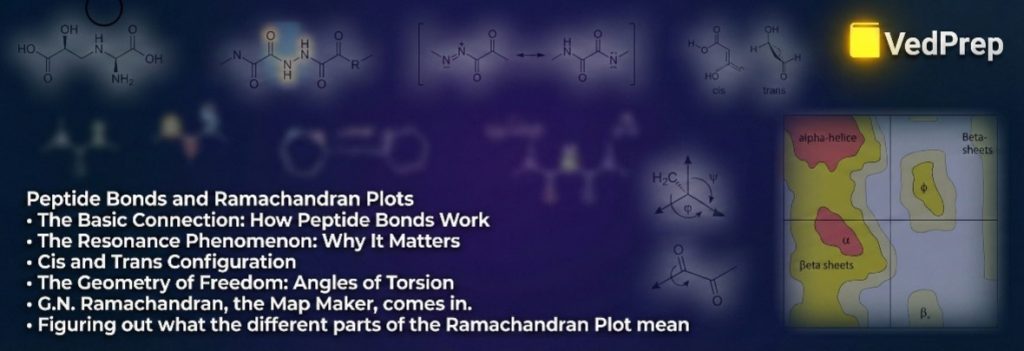
What the Ramachandran Plot Means
The Ramachandran plot is like a 2D graph.
The X-axis shows Phi ($\phi$) values from $-180^\circ$ to $+180^\circ$.
Y-axis: Psi ($\psi$) values from -180° to +180°.
The Landscape: The plot is not filled out evenly. There are certain “islands” in the allowed areas where atoms don’t crash. The rest of the “ocean” is made up of areas where steric clashes happen and are not allowed.
This graph shows the “traffic rules” for how proteins fold. It tells us that Peptide Bonds keep the local structure in place, but the overall 3D shape is only determined by keeping atoms from colliding.
Figuring out what the different parts of the Ramachandran Plot mean
The plot shows three main “Allowed Regions” for a standard L-amino acid, such as Alanine. In the 2026 exams, you will definitely have to find these areas.
1. The Beta-Sheet Area (Top Left)
Coordinates: $\phi \approx -120^\circ$ to $-140^\circ$, and $\psi \approx +110^\circ$ to $+135^\circ$.
Structure: This is the large area in the second quadrant. It is similar to long structures. This is where both parallel and anti-parallel β-sheets fall.
Why here? In this shape, the side chains go up and down, which makes the distance between atoms as big as possible and keeps them from bumping into each other.
2. The Right-Handed Alpha-Helix Area (Bottom Left)
Coordinates: $\phi \approx -60^\circ$, $\psi \approx -45^\circ$.
Structure: This area in the third quadrant is very densely packed.
Why here? This twist lets the Carbonyl Oxygen backbone of residue $i$ make a hydrogen bond with the Amide Hydrogen of residue $i+4$. In biology, it is the most common secondary structure.
3. The Alpha-Helix Region on the Left Hand (Top Right)
Coordinates: $\phi \approx +60^\circ$ and $\psi \approx +45^\circ$.
Structure: In the first quadrant.
The problem is that L-amino acids don’t often make left-handed helices because their side chains get in the way of the backbone. This area doesn’t have many people living in it; it usually only has Glycine residues.
The Outliers: Glycine and Proline
The “Standard” Ramachandran plot works for 18 of the 20 amino acids. The other two, Glycine and Proline, don’t follow the rules.
Glycine: The Acrobat Who Can Move
The side chain of glycine is a hydrogen atom ($R = H$). It is very small.
Effect: Glycine has a lot of freedom to rotate because it doesn’t have a big side chain that could cause steric crashes.
The Plot: The Ramachandran plot for Glycine is nearly symmetrical. It has given areas in all four quadrants.
Biological Role: Glycine is found in places where flexibility is needed, like tight turns and loops. It works like a hinge.
Proline: The Stiff Kink
Proline is an amino acid. Its side chain is connected to its own nitrogen.
The angle $\phi$ is locked at about $-60^\circ$. It can’t turn.
The Plot: The Ramachandran plot for proline is the most limited. It is mostly limited to the Alpha-helical region and the Polyproline helix region.
Biological Role: Proline breaks up helices. It is too stiff to fit comfortably in the middle of a normal $\alpha$-helix.
Peptide Bonds and Technology in 2026
Why are we still so interested in peptide bonds in 2026? Because they are the building blocks of the biotech revolution.
Validation of AI Structures
Scientists use the Ramachandran plot as a “Lie Detector” when AI tools like Alpha Fold make a protein structure. If the AI sees a structure where the $\phi$ and $\psi$ angles are in the “Disallowed Region,” the model is probably wrong. Even a supercomputer can’t break the physical laws that govern steric clashes and the rigidity of peptide bonds.
Peptidomimetics in the Design of Drugs
A lot of modern drugs work like proteins. But digestive enzymes (proteases) can easily break natural peptide bonds. Medicinal chemists created “Peptidomimetics” in 2026. These are molecules that look like Peptide Bonds but have different atoms, like replacing Nitrogen with Carbon.
These altered bonds must still follow certain geometric rules in order to fit into the drug target, which is a viral receptor. Chemists can make these synthetic drugs stay in the “active” shape by knowing the Ramachandran constraints.
Linking the Dots: From Bond to Function
There are a lot of limits that make the journey from the chemistry of peptide bonds to their biological function.
In chemistry, resonance makes peptide bonds flat.
Physics: Steric hindrance makes it hard for $C_\alpha$ to rotate.
In geometry, the Ramachandran plot shows the allowed rotations.
Structure: Only certain shapes, like the $\alpha$-helix and $\beta$-sheet, fit into these allowed areas.
Function: These shapes come together to make the active sites of enzymes or the pockets where antibodies bind.
If the Peptide Bonds lost their partial double-bond character, the Ramachandran plot would go away, the secondary structures would fall apart, and biological function would stop.
Learning Structural Biology with VedPrep
To understand how complex proteins and other molecules are in three dimensions, you need to do more than just read two-dimensional text. You need to be able to see the rotation of the angles $\phi$ and $\psi$, the fact that peptide bonds are planar, and the steric clashes in the Ramachandran plot.
This is where VedPrep changes the way you learn.
We at VedPrep think that you should see structural biology, not just memorize it.
3D Molecular Visualization: Our modules have interactive 3D models that let you rotate Peptide Bonds and see steric clashes happen in real time. You don’t just memorize the “Disallowed Region”; you also understand why atoms crash there.
The Alpha Fold Integration:
We show you how to read modern structural data. Our curriculum links the classic Ramachandran plot to AI predictions from 2026, getting you ready for the questions of the future, not the past.
We look at past CSIR NET and GATE questions to show you the “Trap Zones,” which are places where you might get confused, like when you mix up the coordinates of parallel and anti-parallel beta sheets or the unique plot of Glycine.
Expert Mentorship:
Get advice from PhDs and researchers who use these plots every day to check new drug targets. They connect what you learn in books with what you do in the lab.
VedPrep gives you the visual, structured, and expert-led help you need to make Structural Biology your best unit, whether you’re having trouble with the resonance of peptide bonds or the quadrants of the Ramachandran plot.
In conclusion
The examination of Peptide Bonds and Ramachandran Plots constitutes the investigation of biological limitations. Nature does not have an endless number of options; it follows strict rules of chemistry and physics.
The stability of peptide bonds comes from their partial double-bond character, while the Ramachandran plot’s steric limits show how to fold them.
As we create new enzymes to break down plastic and antibodies to fight future pandemics in 2026, these basic rules will still guide us. We can change biology to fit our needs, but we can’t break the rules of physics that govern it.
For both the student and the scientist, learning these ideas is the first step to understanding the language of life. So, the next time you see a complicated protein structure, take a closer look. Look at the stiff plates of the Peptide Bonds, picture the angles that they turn, and admire the beautiful shapes that keep life going.
Frequently Asked Questions (FAQs)
How are peptide bonds formed in a biological context?
Ans: Peptide bonds are formed through a dehydration synthesis (condensation) reaction where the $\alpha$-carboxyl group of one amino acid joins the $\alpha$-amino group of another, removing a water molecule. In the biological context of 2026, this is viewed as an energy-dependent process carried out by the ribosome.
Why are peptide bonds considered rigid and planar?
Ans: Peptide bonds exhibit "Resonance," where the nitrogen atom's lone pair spreads into the carbonyl group, giving the C-N bond about 40% double-bond character. This partial double bond prevents rotation, locking the six atoms of the peptide unit ($C_\alpha, C, O, N, H, C_\alpha$) into a flat, rigid plane
What is the difference between Cis and Trans configurations, and which is more common?
Ans: In the Trans configuration, the two $\alpha$-carbons are on opposite sides of the bond, which reduces steric hindrance; this occurs in 99.6% of bonds. The Cis configuration places the $\alpha$-carbons on the same side, creating crowding, and is generally rare.
Why is Proline considered an exception to the Trans preference?
Ans:
Proline's side chain loops back to form a ring with its own nitrogen, making the energy difference between Cis and Trans configurations small. Consequently, about 10% of peptide bonds involving Proline are Cis, often creating "kinks" or turns in the protein chain.
What are Torsional Angles (Phi and Psi)?
Ans: These are the points of rotation around the central $\alpha$-carbon ($C_\alpha$). Phi ($\phi$) is the rotation angle between Nitrogen ($N$) and the Alpha Carbon ($C_\alpha$), while Psi ($\psi$) is the rotation angle between the Carbonyl Carbon ($C$) and the Alpha Carbon ($C_\alpha$).
What determines the "Allowed Regions" in a Ramachandran Plot?
Ans: The plot maps possible combinations of $\phi$ and $\psi$ angles based on steric hindrance—treating atoms as hard spheres that cannot occupy the same space. "Allowed regions" are "islands" where atoms don't crash into each other, while the rest indicates forbidden steric clashes.
Where are Beta-Sheets located on the Ramachandran Plot?
Ans: Beta-sheets are found in the large area of the top-left quadrant (second quadrant). The coordinates are approximately $\phi \approx -120^\circ$ to $-140^\circ$ and $\psi \approx +110^\circ$ to $+135^\circ$.
What are the coordinates for a Right-Handed Alpha-Helix?
Ans: Right-handed $\alpha$-helices are located in the densely packed bottom-left quadrant (third quadrant). The typical coordinates are $\phi \approx -60^\circ$ and $\psi \approx -45^\circ$.
Why are Left-Handed Alpha-Helices rare in nature?
Ans: Located in the top-right quadrant (first quadrant), left-handed helices are rare for L-amino acids because their side chains clash with the backbone. This region is typically populated only by Glycine residues.
How does Glycine's behavior on the plot differ from other amino acids?
Ans: Glycine has a small hydrogen atom as a side chain ($R=H$), giving it immense freedom to rotate without steric crashes. Its Ramachandran plot is nearly symmetrical, featuring allowed areas in all four quadrants.