Comprehensive Mastery of the Second Law of Thermodynamics
The Second Law of Thermodynamics states that the total entropy of an isolated system can never decrease over time; it can only remain constant or increase. This fundamental principle dictates the direction of spontaneous processes and establishes that heat cannot naturally flow from a colder body to a hotter body without external work.
Defining the Second Law of Thermodynamics and Entropy
The Second Law of Thermodynamics introduces entropy as a measure of a system’s thermal energy per unit temperature that is unavailable for doing useful work. While the First Law focuses on energy quantity, this law emphasizes energy quality and the inevitable progression toward disorder in every natural transformation.
Entropy is often described as the degree of randomness or chaos within a system. According to the Second Law of Thermodynamics, any spontaneous process in an isolated system leads to an increase in universal entropy. This occurs because energy naturally tends to spread out or disperse rather than stay concentrated. In the study of Physical Chemistry of CUET PG 2026, entropy is treated as a state function, meaning its change depends only on the initial and final states of the substance.
For students preparing for Thermodynamics in CUET PG Exam, understanding entropy calculations is essential. The law implies that while energy is conserved, it becomes less “useful” after every transaction. For example, when energy is converted from chemical to mechanical forms, some is always dissipated as heat. This dissipation ensures that the entropy of the universe is constantly rising, a concept often referred to as the “arrow of time” in physics.
The Kelvin-Planck Statement: Limitations of Heat Engines
The Kelvin-Planck statement of the Second Law of Thermodynamics asserts that it is impossible to construct a device operating in a cycle that produces no effect other than the extraction of heat from a single reservoir and the performance of an equivalent amount of work.
This statement specifically addresses the efficiency limits of heat engines. It confirms that no engine can ever be 100% efficient. A portion of the heat absorbed from a high-temperature source must always be rejected to a lower-temperature sink. This rejected energy is a physical necessity required to complete the thermodynamic cycle. In the Physical Chemistry of CUET PG 2026, this principle explains why perpetual motion machines of the second kind are scientifically impossible.
When tackling Thermodynamics in CUET PG Exam questions, candidates must relate this statement to the efficiency formula. The maximum efficiency is determined by the temperatures of the heat reservoirs rather than the working fluid. Even under ideal conditions, the Second Law of Thermodynamics imposes a boundary on how much thermal energy can be converted into mechanical motion. This makes it a vital topic for understanding power generation and industrial motor design.
The Clausius Statement: Directionality of Heat Flow
The Clausius statement of the Second Law of Thermodynamics specifies that it is impossible to construct a device that operates in a cycle and produces no effect other than the transfer of heat from a cooler body to a warmer body.
This version of the law defines the natural direction of heat transfer. Spontaneous heat flow always moves from high temperature to low temperature. To reverse this flow, such as in a refrigerator or air conditioner, an external energy source must perform work on the system. The Clausius statement is a foundational concept in Physical Chemistry of CUET PG 2026, providing the theoretical framework for refrigeration cycles and heat pumps.
For those focusing on Thermodynamics in CUET PG Exam, the Clausius statement helps clarify the concept of “unnatural” processes. While the First Law does not forbid heat flowing from cold to hot, the Second Law of Thermodynamics identifies such an event as a violation of entropy requirements. By requiring work input for cooling, the law ensures that the total entropy of the system and its surroundings still increases, even if the local entropy of the refrigerated space decreases.
Carnot’s Theorem and Ideal Efficiency
Carnot’s Theorem, derived from the Second Law of Thermodynamics, establishes that no heat engine operating between two thermal reservoirs can be more efficient than a reversible Carnot engine operating between those same reservoirs. It further states that all reversible engines have the same efficiency regardless of the working substance.
The Carnot cycle serves as the ultimate benchmark for thermal systems. It consists of two isothermal and two adiabatic processes. Because the Second Law of Thermodynamics limits the conversion of heat to work, the Carnot efficiency depends solely on the absolute temperatures of the source (TH) and sink (TL). This relationship is a high-yield area for Physical Chemistry of CUET PG 2026 aspirants, as it connects theoretical limits with practical engineering constraints.
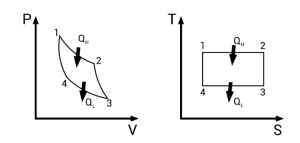
Mastering the Carnot cycle is indispensable for Thermodynamics in CUET PG Exam success. It demonstrates that as the temperature difference between the source and sink increases, the potential for efficiency also grows. However, since reaching absolute zero for a sink is impossible (as per the Third Law), the Second Law of Thermodynamics ensures that perfectly efficient energy conversion remains a theoretical ideal rather than a reachable reality.
Statistical Interpretation of the Second Law
From a statistical mechanics perspective, the Second Law of Thermodynamics is a statement of probability. It suggests that a system will most likely evolve from a state of lower probability (ordered) to a state of higher probability (disordered) because there are many more ways to be disordered than ordered.
Ludwig Boltzmann quantified this by relating entropy to the number of microstates (W) through the formula S = k \ln W. In an isolated system, the molecules move toward arrangements that maximize the number of possible configurations. This statistical approach is a core part of the Physical Chemistry of CUET PG 2026, bridging the gap between microscopic particle behavior and macroscopic thermodynamic observations.
In the Thermodynamics in CUET PG Exam, students are often asked to evaluate entropy changes based on molecular arrangements. For instance, when a gas expands into a vacuum, the number of available positions for each molecule increases, leading to a rise in microstates and, consequently, a rise in entropy. The Second Law of Thermodynamics provides the macro-scale validation for these micro-scale movements, confirming that the universe favors states of maximum statistical likelihood.
Spontaneity and Gibbs Free Energy
The Second Law of Thermodynamics provides the criteria for chemical spontaneity through the concept of Gibbs Free Energy (G). For a process to be spontaneous at constant temperature and pressure, the change in Gibbs Free Energy must be negative (Δ G < 0).
Δ G integrates the system’s enthalpy change with its entropy change (Δ G = \Δ H – T Δ S). If the Second Law of Thermodynamics requires the total entropy of the universe to increase, then $\Delta G$ essentially tracks this requirement from the perspective of the system alone. This makes it the most practical tool in the Physical Chemistry of CUET PG 2026 for predicting whether a chemical reaction will proceed without external intervention.
During the Thermodynamics in CUET PG Exam, candidates must analyze the interplay between enthalpy and entropy. A reaction might be endothermic (absorbing heat) but still spontaneous if the increase in entropy is large enough to overcome the enthalpy barrier at high temperatures. The Second Law of Thermodynamics ensures that the “energy tax” paid to entropy determines the final direction of chemical equilibrium.
Practical Application: Industrial Refrigeration and HVAC
Refrigeration systems are practical applications of the Clausius statement of the Second Law of Thermodynamics. These systems use mechanical work to extract heat from a low-temperature environment and discharge it into a higher-temperature surroundings, effectively moving energy against its natural gradient.
In a standard refrigeration cycle, a refrigerant absorbs heat while evaporating at low pressure and releases heat while condensing at high pressure. This process requires a compressor, which provides the work input mandated by the Second Law of Thermodynamics. Without this work, heat would never move out of the cold storage area. This application is a major theme in the Physical Chemistry of CUET PG 2026, as it involves phase changes and heat transfer coefficients.
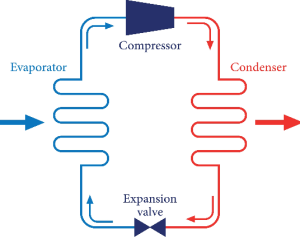
For students of Thermodynamics in CUET PG Exam, understanding the Coefficient of Performance (COP) is essential. The COP measures the effectiveness of a refrigerator or heat pump. The Second Law of Thermodynamics limits the maximum possible COP, mirroring the efficiency limits of heat engines. Studying these systems helps students visualize how abstract laws govern the technology used in everyday climate control and food preservation.
Critical Perspective: The “Heat Death” of the Universe
A common philosophical and scientific extension of the Second Law of Thermodynamics is the “Heat Death” theory. This hypothesis suggests that since the entropy of the universe is always increasing, the universe will eventually reach a state of maximum entropy. At this point, all energy will be distributed uniformly, no gradients will exist, and no more work can be performed, resulting in a cold, dark, and lifeless cosmos.
However, this perspective is often criticized for applying equilibrium thermodynamics to the entire universe, which may not be a closed or simple system. Some cosmological models, including those involving dark energy or general relativity, suggest that the expansion of space might outpace the production of entropy, or that gravity creates new pockets of low entropy. For those studying Physical Chemistry of CUET PG 2026, it is important to recognize that while the Second Law of Thermodynamics is absolute in local engineering systems, its application to the infinite scale of the universe remains a subject of intense debate.
Entropy Changes in Reversible and Irreversible Processes
In a reversible process, the total entropy of the universe remains constant because the system and surroundings can return to their original states without leaving any change. In an irreversible process, which characterizes all real-world events, the total entropy of the universe always increases.
The distinction between reversible and irreversible paths is a frequent point of assessment in Thermodynamics in CUET PG Exam. Friction, turbulence, and rapid expansion are all sources of irreversibility that generate “extra” entropy. In the Physical Chemistry of CUET PG 2026, students learn that while we use reversible paths to calculate entropy changes (dS = dQ/T), real processes always produce more entropy than the ideal calculation suggests.
By understanding this gap, researchers can improve the efficiency of industrial processes. Reducing irreversibility—such as by using better lubricants or slower expansion steps—minimizes entropy production. The Second Law of Thermodynamics serves as a guide for sustainable engineering, reminding us that minimizing entropy generation is the key to preserving the useful energy available in our resources.
Second Law in Biological Systems
Biological organisms appear to defy the Second Law of Thermodynamics by creating highly ordered structures from disordered raw materials. However, they maintain this internal order by exporting a greater amount of entropy to their surroundings through heat and metabolic waste.
Life is not a violation of thermodynamics but a sophisticated management of it. Plants capture low-entropy sunlight and convert it into high-entropy heat while building complex molecules. Animals consume these molecules and release heat and gases. This exchange ensures that while the organism stays ordered, the “total” entropy of the biosphere and the sun still increases. This concept is vital for the Physical Chemistry of CUET PG 2026, specifically in the study of bioenergetics.
For candidates of Thermodynamics in CUET PG Exam, this illustrates the importance of the “system vs. surroundings” boundary. You can decrease the entropy of a system (like a growing cell) as long as you increase the entropy of the surroundings by a larger margin. The Second Law of Thermodynamics governs the energy budget of every living cell, dictating the efficiency of ATP synthesis and the limits of muscular work.
Calculating Entropy for Different Transformations
To calculate entropy changes for various substances, chemists integrate the ratio of heat transfer to temperature. For ideal gases, the entropy change depends on both volume and temperature, while for phase changes, it is simply the latent heat divided by the transition temperature.
Standard formulas used in Physical Chemistry of CUET PG 2026 include:
- Temperature Change: Δ S = nCp ln (T2/T1)
- Volume Change: Δ S = nR ln (V2/V1)
- Phase Change: Δ S = Δ H fusion / T melting
The Second Law of Thermodynamics ensures that these values are consistently applied to predict reaction feasibility. In the Thermodynamics in CUET PG Exam, students must be proficient in combining these formulas for multi-step processes. For example, calculating the entropy change when ice at -10°C is converted to steam at 110°C requires five distinct steps, each obeying the core principles of entropy accumulation.
Exam Strategy for Physical Chemistry of CUET PG 2026
Success in the Physical Chemistry of CUET PG 2026 requires a balanced approach to the Second Law of Thermodynamics, focusing on both conceptual statements and rigorous mathematical problem-solving.
- Focus on Statements: Be prepared for “Which of the following is true” questions regarding Kelvin-Planck and Clausius statements.
- Master the Math: Practice Δ G and Δ S calculations for varied conditions (isobaric, isochoric, and isothermal).
- Interpret Graphs: Understand how entropy changes on T-S (Temperature-Entropy) diagrams, especially for the Carnot cycle.
Regularly solving previous years’ papers for Thermodynamics in CUET PG Exam will reveal that entropy and spontaneity are among the most heavily weighted topics. A deep grasp of the Second Law of Thermodynamics not only ensures marks in the chemistry section but also provides the logical framework needed for advanced physics and biochemistry modules.
Summary of Thermodynamic Spontaneity
The Second Law of Thermodynamics is the final arbiter of what can and cannot happen in the physical world. It transforms the study of energy from a simple balance sheet into a complex map of possibility. By defining the limits of efficiency and the direction of time, it provides the essential context for all chemical and physical changes.
Whether you are calculating the efficiency of a power plant or the spontaneity of a lab-scale reaction, the Second Law of Thermodynamics remains your primary guide. For the Physical Chemistry of CUET PG 2026, this law is the bridge between the laws of motion and the realities of energy dissipation. Mastering it is not just an academic requirement for Thermodynamics in CUET PG Exam—it is a prerequisite for understanding the fundamental behavior of the universe.
Would you like me to generate a table comparing the efficiencies of different thermodynamic cycles to help with your CUET PG 2026 preparation?
For further updates and notifications visit the official website.
| Related Link |
| Third Law of Thermodynamics |










