Bohr’s Theory accounts for the structural stability of atoms by suggesting that electrons travel in definite circular paths around the nucleus. This framework brought in the concept of quantized angular momentum and established discrete energy states for the hydrogen atom. It accurately computes the Rydberg constant and clarifies the distinct line spectra observed in single-electron systems.
Fundamental Bohr’s Atomic Model Postulates
Bohr’s Atomic Model Postulates lay the groundwork for grasping atomic structure within the IIT JAM Biotechnology Syllabus. These core ideas shift chemistry from a classical physics framework toward quantum mechanics by outlining electron conduct inside an atom. The postulates specify the precise orbits and energy levels that ensure an atom’s enduring stability.
Electrons revolve around the nucleus in specific circular paths called orbits or stationary states. Each orbit possesses a definite amount of energy. An electron in a particular orbit does not radiate energy while moving. This explains why atoms do not collapse despite the electromagnetic attraction between the nucleus and electrons.
An electron’s energy is stable while it remains in its allotted path. Shifts in energy only happen when an electron transitions between distinct tiers. Taking in energy promotes an electron to an upper level. Releasing energy takes place when an electron drops to a lesser level. This alteration in energy matches the gap between the two particular tiers.
Quantization of Angular Momentum in Atomic Orbits
Quantization of Angular Momentum marks the biggest break from classical physics within Bohr’s Theory. Bohr suggested that an electron is restricted to orbits where its angular momentum is a whole-number multiple of a defined constant. This constraint guarantees that solely particular orbital sizes are physically allowed for an electron.
The mathematical expression for this condition involves the mass of the electron, its velocity, and the radius of the orbit. According to Bohr, the angular momentum L follows the rule:
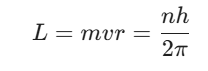
In this expression, n is a positive integer known as the principal quantum number. The variable h represents Planck’s constant. Because n must be an integer like 1, 2, or 3, the angular momentum is quantized. This quantization prevents the electron from existing at arbitrary distances from the nucleus. It forces the electron into discrete shells labeled K, L, M, and N.
Energy Levels of Hydrogen Atom and Spectral Lines
The quantized energy states of a hydrogen atom depict the measured distribution of energy inside a solitary electron system. These states dictate the light’s frequency and wavelength when the atom either radiates or takes in energy. Grasping these levels is crucial for proficiency in the periodicity of properties segment found within the IIT JAM Biotechnology Syllabus.
Bohr’s Theory allows for the calculation of the energy associated with each shell. The energy of an electron in the n-th orbit is given by:
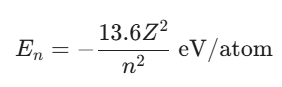
For a hydrogen atom, the atomic number Z is 1. As n increases, the energy becomes less negative, meaning the electron is less tightly bound to the nucleus. The ground state corresponds to n = 1, where the electron has the lowest possible energy. Excited states occur at higher n values. When an electron transitions from a higher energy level E2 to a lower level E1, it emits a photon with energy ΔE = E2 – E1.
Mathematical Expressions in Bohr’s Theory
Determining values for atomic radius, speed, and power is crucial for tackling quantitative questions frequently encountered in competitive assessments. Bohr’s Theory offers exact equations resulting from the equilibrium between electrical attraction and the need for circular motion. These expressions apply strictly to hydrogen and hydrogen like ions such as He+, Li2+, and Be3+.
The following table summarizes the primary mathematical expressions used in Bohr’s Theory:
| Parameter | Formula Expression | Relationship with n and Z |
|---|---|---|
| Radius of n-th Orbit (rn) | rn = 0.529 ×(n2⁄Z) Å | r ∝n2 / Z |
| Velocity of Electron (vn) | vn = 2.18 ×106 × Z/n m/s | v ∝ Z / n |
| Total Energy (En) | En = -13.6 × Z2/n2 eV | E ∝ -Z2 / n2 |
| Frequency of Radiation (ν) | ν = ΔE/h | ν = RH c (1/n12– 1/n22) |
These equations enable learners to foresee how electrons will act in various quantum states. For example, boosting the principal quantum number ‘n’ substantially expands the orbital size but lessens the orbital speed.
Quantitative Analysis of Hydrogen Spectral Series
Bohr’s Theory effectively accounts for the hydrogen spectrum by describing electron movement between energy states. Each set of spectral lines relates to shifts concluding at a particular lower energy state. This capacity for forecasting offered initial proof that Bohr’s concept surpassed earlier atomic ideas.
The wavelength of emitted light is calculated using the Rydberg formula:
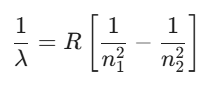
The table below details the different spectral series identified in the hydrogen atom:
| Series Name | Final Level (n1) | Initial Level (n2) | Spectral Region |
|---|---|---|---|
| Lyman | 1 | 2, 3, 4… | Ultraviolet |
| Balmer | 2 | 3, 4, 5… | Visible |
| Paschen | 3 | 4, 5, 6… | Near Infrared |
| Brackett | 4 | 5, 6, 7… | Infrared |
| Pfund | 5 | 6, 7, 8… | Far Infrared |
The Balmer collection of lines holds special significance since its markings lie inside the visible light range. Researchers are able to view these lines firsthand employing a spectroscope. The Lyman set features the most energetic shifts, owing to the electron dropping down to the base energy level.
Application of Bohr’s Theory in Biotechnology
Bohr’s Theory, while based on chemical concepts, has significance within the IIT JAM Biotechnology syllabus. The composition of atoms dictates the chemical characteristics of materials essential for life. Understanding energy levels is key for investigating processes such as light emission (fluorescence, phosphorescence) and light quantification (spectrophotometry) seen in the lab.
In the realm of biotechnology, numerous analytical methods are dependent on electronic shifts. For instance, spectrophotometry in the UV-Vis range quantifies how molecules take in illumination, a process that includes moving electrons across distinct energy states. The notion of separate energy steps explains why life’s pigments, like chlorophyll or heme, take in specific light wavelengths. Lacking the foundation established by Bohr’s Theory, advances in contemporary imaging methods such as X-ray diffraction analysis or magnetic resonance would not have materialized.
Bohr’s Theory Limitations and Theoretical Failures
The chief limitations of Bohr’s Theory indicate the need for a more advanced quantum structure that ultimately succeeded it. While Bohr’s depiction was an improvement upon Rutherford’s idea, his construct could not explain complex atomic phenomena. These deficiencies stem from the attempt to apply tenets of classical mechanics to quantum objects.
Bohr’s Theory fails to explain the observed spectra in atoms containing more than one electron. Although accurate for hydrogen, it cannot successfully predict the spectral lines for helium or more intricate atoms. Furthermore, the theory overlooks the electron’s wave-like properties, a concept later established by de Broglie. Bohr conceptualized the electron strictly as a particle following a definite trajectory.
Critical Perspective on the Circular Orbit Assumption
It is often thought that Bohr’s circular paths for electrons are physically real. Nevertheless, this notion is deeply mistaken. Contemporary quantum theory reveals that electrons do not trace out trajectories. Instead, they reside in three-dimensional areas termed orbitals where their presence is most likely.
The circular orbit model is a useful simplification for teaching, but it fails in high precision chemistry. You must recognize that Bohr’s orbits are actually projections of energy states. If you rely solely on the circular path concept, you cannot understand the complex shapes of p, d, and f orbitals. To mitigate this limitation, treat Bohr’s Theory as a stepping stone. Use it to understand energy quantization, but switch to the Schrödinger wave equation for describing electron spatial distribution.
Practical Example: Calculating Ionization Energy
Determining ionization energy offers a useful application of Bohr’s Model for test readiness. Ionization energy is the energy needed to completely extract an electron from an atom in its fundamental state. For a hydrogen atom, this means moving the electron from n = 1 to n = ∞.
Using the energy formula:
E1 = -13.6 eV
E∞ = 0 eV
ΔE = E∞ – E1 = 0 – (-13.6) = 13.6 eV
If you apply this to a He+ ion, the Z value becomes 2. The ionization energy increases fourfold because E is proportional to Z2.
E1(He+) = -13.6 × 22 = -54.4 eV
This higher energy requirement explains why it is harder to remove electrons from ions with higher nuclear charges. This logic helps you predict periodicity in properties across the periodic table.
Conclusion of Atomic Properties and Trends
In the IIT JAM Biotechnology Syllabus, these trends explain the reactivity of metal ions in enzyme catalysis. The energy required to shift or remove an electron determines how a molecule will interact with biological substrates. Bohr’s work ensures that you can quantify these interactions rather than merely describing them. VedPrep offers specialized courses to cover complex subject areas like Bohr’s Theory for giving a clear concept.
Frequently Asked Questions (FAQs)
What is Bohr’s Theory of the atom?
Bohr’s Theory describes the atom as a small, positively charged nucleus surrounded by electrons traveling in circular orbits. This model suggests that electrons occupy fixed positions with specific energy levels. It prevents the atom from collapsing by stating that electrons do not radiate energy while staying in their designated stationary orbits.
What are the primary Bohr’s Atomic Model Postulates?
The postulates state that electrons move in discrete circular orbits. Angular momentum is quantized and equals an integral multiple of h/2π. Electrons only change energy by jumping between these orbits. Energy is emitted when an electron drops to a lower shell and absorbed when it moves to a higher shell.
How does Bohr’s Theory explain atomic stability?
Classical physics predicted that accelerating electrons would lose energy and spiral into the nucleus. Bohr resolved this by proposing stationary states. In these states, an electron maintains constant energy. The electron remains stable because it only loses energy during a specific transition to a lower level, not during continuous orbital motion.
What is the significance of the principal quantum number n?
The principal quantum number $n$ defines the shell or energy level of an electron. It starts from 1 for the shell closest to the nucleus, known as the K shell. As $n$ increases, the distance of the electron from the nucleus and its total energy also increase.
Which atoms does Bohr’s Theory apply to?
Bohr’s Theory applies to hydrogen and hydrogen like species. These are single electron systems such as He+, Li2+, and Be3+. The model accurately predicts the spectral lines for these ions but fails for any system containing two or more electrons due to inter-electronic repulsions.
What is the ground state of an atom in Bohr’s Model?
The ground state represents the lowest energy level of an atom. For hydrogen, this occurs at n=1. At this level, the electron is most tightly bound to the nucleus. Any energy state higher than n=1 is an excited state where the electron is less stable.
Why did Bohr’s Theory need the Quantization of Angular Momentum?
Quantization was necessary to explain why only specific orbits are allowed. Without it, electrons could exist at any distance from the nucleus. By requiring angular momentum to be nh/2π, Bohr restricted electrons to paths where their wave properties would naturally reinforce themselves, creating stable energy states.
Why does Bohr’s Theory fail for helium atoms?
Helium has two electrons. Bohr’s model only considers the attraction between one electron and the nucleus. It ignores the repulsion between the two electrons. This additional force changes the energy levels significantly, making Bohr’s simple calculations inaccurate for any multi electron system.
How does Bohr’s Theory relate to the IIT JAM Biotechnology Syllabus?
The IIT JAM Biotechnology Syllabus includes the structure and properties of atoms as a core chemistry component. Candidates must understand Bohr’s Theory to master periodicity and electronic transitions. These concepts are fundamental for practical biotechnology techniques like spectrophotometry and molecular binding studies.
What is the difference between an orbit and an orbital?
An orbit is a well defined circular path used in Bohr’s Theory. An orbital is a three dimensional region where the probability of finding an electron is high. Modern quantum mechanics uses orbitals because the Heisenberg Uncertainty Principle proves that precise circular orbits cannot exist.
What happens to an electron at n = infinity?
When $n$ reaches infinity, the radius becomes infinite and the energy becomes zero. This means the electron is no longer under the influence of the nucleus. The atom is ionized. The energy required to reach this state from n=1 is the ionization energy
What are the Bohr’s Theory Limitations regarding spectral fine structure?
High resolution spectroscopes show that single spectral lines are actually composed of multiple closely spaced lines. Bohr’s Theory cannot explain this fine structure. It assumes all electrons in a main shell have the same energy, ignoring the existence of subshells like s, p, d, and f.
How do the Zeeman and Stark effects disprove Bohr’s model?
The Zeeman effect occurs in magnetic fields and the Stark effect in electric fields. Both cause spectral lines to split into further components. Bohr’s Theory provides no mechanism for this splitting. This suggests that the orientation of electron paths in space is more complex than simple circular orbits.
How does the model explain the Lyman series?
The Lyman series occurs when electrons fall from any higher orbit (n=2, 3, 4...) to the first orbit (n=1). These transitions involve the largest energy changes. Consequently, the emitted radiation falls in the ultraviolet region of the electromagnetic spectrum.












