Collision theory For IIT JAM is a fundamental concept in physical chemistry that explains the rate of chemical reactions based on the collisions of reacting molecules, requiring sufficient energy for successful outcomes.
Collision Theory Syllabus For IIT JAM – Key Textbooks
The topic of Collision theory belongs right in Unit 2.8: Chemical Kinetics of the official IIT JAM Chemistry syllabus. This unit bridges the gap between thermodynamics (will a reaction happen?) and kinetics (how fast will it happen?).
Chemical kinetics is a major scoring area in this unit. It zeroes in on reaction rates, rate laws, and rate constants. To ace these questions, you need to understand how Collision theory serves as the backbone for determining reaction rates.
For a solid, deep-dive study, you can rely on standard textbooks like:
-
Physical Chemistry by P.W. Atkins: This is the gold standard. It gives you a comprehensive look at physical chemistry concepts, especially chemical kinetics and Collision theory.
-
Principles of Physical Chemistry by Puri, Sharma, & Pathania: A favorite among Indian aspirants for its structured numerical approach.
There are no official weightage percentages tied to this specific topic, but ignoring it is a bad idea. A rock-solid foundation here makes scoring in kinetics and thermodynamics much easier on exam day.
Understanding Collision Theory For IIT JAM – Main Concept Explanation
Let’s break down the main concept. Collision theory states that reaction rates depend entirely on two things: how often reactant molecules collide, and how effective those collisions actually are. Think of it like trying to crack open a walnut. If you tap it gently, nothing happens. If you smash it with the right force, it breaks. The rate of a chemical reaction is simply the number of these successful molecular smashes happening per second.
For a collision to be successful, the molecules must bring enough kinetic energy to the table to break existing chemical bonds and form new ones. This energy threshold is the activation energy ($E_a$). It is the minimum energy barrier molecules must clear. If they don’t have it, they just bounce apart completely unchanged.
According to Collision theory, two primary factors control the reaction rate:
-
Collision frequency: The total number of collisions happening per unit of time.
-
Collision energy: The actual energy that the colliding molecules pack.
These factors work together to lock in the overall rate. When you are prepping for the IIT JAM and similar exams, mastering the math between reaction rates, collision frequency, and activation energy is non-negotiable. Once you picture this at the molecular level, solving kinetics problems becomes second nature.
Worked Example
Let’s look at a typical problem you might see in the exam.
Part A: The rate constant (k) for a certain reaction is 2.5 s-1 at 25°C. If the concentration of reactants is 0.5 M, calculate the rate of reaction, assuming a first-order reaction.
The rate law for a first-order reaction is:
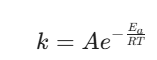
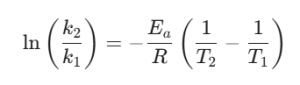

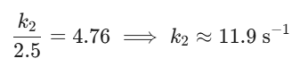
Real-World Applications
To make Collision theory stick, it helps to see how it operates outside the textbook. Take a basic combustion reaction, like burning firewood or gasoline. The fuel molecules have to slam into oxygen molecules. If you crank up the heat or compress the gases, you jumpstart the collision frequency and energy, causing the fire to burn much faster.
Another great example is the catalytic converter in your car. It is designed to clean up toxic exhaust gases before they leave the tailpipe. Normally, converting these harmful gases requires a massive amount of energy. The catalyst inside provides a clever shortcut by lowering the activation energy barrier. Because the energy hurdle is lower, a much higher percentage of daily molecular collisions turn into successful reactions, turning pollutants into safer gases.
We also use this theory to scale up industrial chemical manufacturing. Think about the Haber-Bosch process, which is used to make ammonia for fertilizers.
| Temperature (°C) | Reaction Rate (mol/s) |
| 500 | 0.05 |
| 600 | 0.15 |
| 700 | 0.30 |
As you can see in this sample data table, raising the temperature causes the reaction rate to climb steadily. Chemical engineers use these trends to find the perfect sweet spot for temperature and pressure, ensuring factories run efficiently without wasting energy.
Exam Strategy – Studying Collision Theory For IIT JAM
When you sit down to study this topic for the JAM, don’t just memorize the postulates and move on. You need to understand the moving parts: how activation energy acts as a gatekeeper, and how the collision frequency factor sets the speed limit.
At VedPrep, we often tell students that the secret to physical chemistry is in the math practice. You want to practice numerical problems until you can handle them in your sleep. Go through previous years’ question papers and tackle timed mock tests. Make sure to compare Collision theory side-by-side with the Arrhenius equation and Transition State Theory. Seeing how these concepts connect will give you a massive advantage when the exam presents tricky, conceptual questions.
Limitations of Collision Theory – Implications For IIT JAM
As helpful as it is, Collision theory isn’t perfect. A major flaw is that it treats complex, organic molecules as simple, hard spheres—like billiard balls. In the real world, shape matters.
Imagine a fictional scenario where Molecule A needs to attach its oxygen end specifically to the nitrogen end of Molecule B. If Molecule A comes flying in backward and hits the wrong side, nothing happens, no matter how much energy it has. The theory completely overlooks this molecular orientation factor.
Because of these gaps, the scientific community developed Transition State Theory (TST). TST offers a much better explanation by focusing on the formation of an “activated complex”—a temporary, high-energy arrangement where old bonds are halfway broken and new ones are halfway formed.
Even with its simplifications, Collision theory remains a foundational pillar of kinetics. It is exactly why examiners love testing you on it in competitive exams like CSIR NET and IIT JAM.
Final Thoughts
Mastering Collision theory isn’t about memorizing a checklist of rules for your exam notes. It is about building an intuition for how the physical world interacts at a microscopic level. For anyone aiming for IIT JAM 2027, connecting theoretical collision rates with practical Arrhenius calculations is your ticket to clean, high-scoring numerical marks in Chemical Kinetics.
If you want to sharpen your prep and get some expert-led guidance, the team at VedPrep has put together comprehensive resources and strategies to help you tackle the exam with confidence. Just remember as you study: every successful reaction—much like your exam preparation—requires the right orientation and exactly the right amount of energy.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
What are the key factors in collision theory?
The key factors are: (1) effective collisions must occur, (2) collisions must have sufficient energy (activation energy), and (3) proper molecular orientation during collision. These factors influence the rate of a chemical reaction.
How does temperature affect collision theory?
Temperature affects collision theory by increasing the kinetic energy of molecules, leading to more frequent and more energetic collisions. This generally increases the rate of reaction as temperature rises.
What is activation energy in collision theory?
Activation energy is the minimum energy required for a collision to be effective and result in a chemical reaction. It is a critical concept in understanding reaction rates and is often represented in energy profiles of reactions.
How does concentration affect collision theory?
Concentration affects collision theory by increasing the frequency of collisions. Higher concentrations mean more molecules in a given space, increasing the likelihood of collisions and, consequently, the rate of reaction.
What is the role of orientation in collision theory?
Orientation plays a crucial role in collision theory as it determines whether a collision is effective. Molecules must be oriented in a specific way for a reaction to occur, adding a steric component to the theory.
How does surface area affect collision theory?
Surface area affects collision theory by providing more sites for collisions to occur. Increased surface area can lead to more collisions and, consequently, a higher rate of reaction, especially in heterogeneous reactions.
What is the significance of effective collisions in collision theory?
Effective collisions are significant because they lead to chemical reactions. For a collision to be effective, the colliding molecules must have sufficient energy (activation energy) and proper orientation.
What are the assumptions of collision theory?
The assumptions include that molecules are hard spheres, all collisions are effective if they have sufficient energy and proper orientation, and the reaction rate is directly proportional to the concentration of reactants.
How can collision theory be applied to IIT JAM Physical Chemistry questions?
Collision theory can be applied to understand and predict the rates of chemical reactions, which is crucial for solving problems related to chemical kinetics in IIT JAM Physical Chemistry. It helps in analyzing how different factors affect reaction rates.
What types of questions about collision theory can be expected in IIT JAM?
Expect questions that relate collision theory to reaction rates, activation energy, and the effects of concentration, temperature, and surface area on reactions. Also, questions may involve applying the theory to specific chemical reactions or kinetics problems.
Can collision theory be used to explain the rate of reaction in industrial processes?
Yes, collision theory can be applied to industrial processes to understand and optimize reaction conditions for higher efficiency and yield. It helps in designing reactors and determining the optimal conditions for reactions.
How can understanding collision theory help in optimizing chemical reactions?
Understanding collision theory helps in optimizing chemical reactions by identifying the conditions that maximize the frequency and effectiveness of collisions, such as adjusting temperature, concentration, and pressure.
What are common mistakes in applying collision theory?
Common mistakes include neglecting the importance of molecular orientation, misinterpreting the role of activation energy, and not accounting for the effects of concentration and temperature on reaction rates. Overlooking the steric factor is also a frequent error.
How can students avoid mistakes in collision theory problems?
To avoid mistakes, students should carefully consider all factors affecting reaction rates, accurately calculate activation energy, and properly apply the concepts to different reaction scenarios. Practice with a variety of problems is essential.










