First law of thermodynamics For IIT JAM states that energy cannot be created or destroyed, only converted from one form to another. This fundamental principle is crucial for solving thermodynamics problems in IIT JAM exams.
Syllabus – Thermodynamics
Thermodynamics isn’t just a chapter; it’s a massive chunk of the IIT JAM Physics syllabus. Usually tucked into the kinetic theory and thermodynamics section, it’s a high-yield area. While legends like Halliday-Resnick-Walker or Knight are great for building a deep foundation, remember that the JAM exam loves to test how you apply these concepts such as First law of thermodynamics to real systems.
At VedPrep, we’ve noticed that students who master the First law of thermodynamics early on find it much easier to tackle tougher topics like entropy or heat engines later. Whether you’re also eyeing GATE or CUET PG, this is the ground floor of your preparation.
First Law of Thermodynamics: A Fundamental Principle
Think of the First Law as a bank account for energy. If you deposit some “cash” (heat, Q) and the system spends some on “shopping” (work, W), whatever is left over stays in the “savings account” (internal energy, ΔU).
The math is simple: ΔU = Q – W
In physics, we usually say W is the work done by the system. If the system expands, it’s doing work (spending energy), so we subtract it. If you compress it, you’re doing work on it, and that internal energy goes up.
Worked Example: Thermodynamic Process
Let’s look at a quick problem on First law of thermodynamics. Say you have a gas in a cylinder. Its internal energy starts at 500 J and ends up at 700 J after a process. During this time, it sucks up 300 J of heat. How much work did the gas actually do?
-
Find the change: ΔU = Ufinal – Uinitial = 700 J – 500 J = 200 J.
-
Plug it in: 200 J = 300 J – W.
-
Solve: W = 100 J.
Therefore, the work done by the system during this thermodynamic process is 100 J.
Common Misconception: Energy Conservation
A common trap is thinking “conservation” means nothing changes. That’s not true. Imagine a fictional scenario where you’re rubbing your hands together on a cold morning. You’re putting in mechanical work, which turns into heat. The total energy in that little “hand-system” is conserved, but the type of energy is flipping from motion to thermal.
- Initial energy: Electrical energy
- Final energy: Mechanical energy
- Total energy: Remains constant
As per First law of thermodynamics, the ΔE = Q – W equation illustrates this concept, where ΔE is the change in energy, Q is the heat added, and W is the work done. This equation shows that energy is conserved, but its form can change. A clear understanding of this concept is essential for solving problems in thermodynamics.
First Law of Thermodynamics For IIT JAM: Applications
This law isn’t just for passing exams; it runs the world. Take a power plant: you burn coal (heat), which makes steam spin a turbine (work). The First law of thermodynamics helps engineers figure out exactly how much electricity they can get out of a ton of fuel.
Even your fridge uses this. It basically works the equation backward by using electricity (work) to move heat from the inside to the outside. At VedPrep, we suggest visualizing these machines when you see a cycle on a P-V diagram. It makes the abstract math feel a lot more “real.”
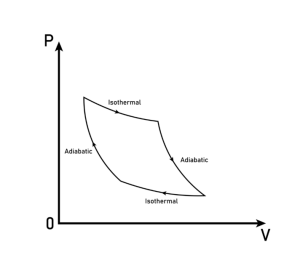
Thermodynamic cycles, such as the Carnot cycle, Otto cycle, and Rankine cycle, are also crucial applications of the first law. These cycles describe the conversion of heat energy into work energy in various engines and power generation systems. They operate under specific constraints, like adiabatic processes and isobaric expansions, to optimize energy conversion efficiency. Understanding these cycles is essential for designing efficient power generation and propulsion systems.
Exam Strategy: Thermodynamics Tips
When you’re staring at a JAM paper, time is your biggest enemy. Here are a few tips to stay ahead:
-
Watch the units: Don’t mix Joules and calories. It sounds basic, but under exam pressure, it happens to the best of us.
-
Identify the process: Is it isothermal (ΔT = 0)? Is it Adiabatic (Q = 0)? Knowing this tells you which part of the ΔU = Q – W equation becomes zero or changes.
-
Practice the graphs: Learn to read $P-V$ diagrams like a pro. The area under the curve is your work done.
For comprehensive and effective preparation, utilizing VedPrep’s study materials for thermodynamics can be highly beneficial. VedPrep offers expert guidance through detailed notes, practice questions, and mock tests tailored to the needs of CSIR NET, IIT JAM, and GATE aspirants. By focusing on understanding thermodynamic processes, practicing problem-solving, and leveraging resources like VedPrep, students can enhance their grasp of thermodynamics and perform confidently in their exams.
First law of thermodynamics For IIT JAM: Important Subtopics
To really crush this section, keep a close eye on these:
-
Internal Energy (U): For an ideal gas, this only depends on temperature. If T doesn’t change, ΔU is zero.
-
Enthalpy (H): Think of this as the “total heat content,” especially useful when pressure is constant.
-
Specific Heat: Know the difference between Cp and Cv. They are the “scaling factors” for how much heat it takes to raise the temperature.
Real-World Example: Thermodynamic Systems
Let’s imagine a fictional gadget—let’s call it the “Super-Pump.” If you pump air into a bike tire really fast, the pump gets hot. Why? You’re doing work so quickly that the heat doesn’t have time to escape (an adiabatic process). That work you did had to go somewhere, so it bumped up the internal energy, which we feel as heat.
This is the same reason why a car engine gets hot. It’s not just the fire; it’s the rapid compression of gases. Understanding these little scenarios makes the First law of thermodynamics feel less like a dry formula and more like a rulebook for how stuff works.
- Power plants utilize thermodynamic cycles, such as the Rankine cycle, to convert heat energy into electrical energy.
- These cycles operate under specific constraints, including temperature and pressure limits.
The study of thermodynamic systems is essential for optimizing energy conversion and utilization in various industries. By understanding the fundamental principles of the First law of thermodynamics, researchers and engineers can develop more efficient and sustainable technologies.
Final Thoughts
At the end of the day, the First law of thermodynamics is just about keeping a good set of books. Energy comes in, energy goes out, and whatever is left stays inside. If you can keep track of those three things, you’re already halfway to a great JAM score.
Keep practicing those P-V cycles and don’t let the sign conventions trip you up. We’re all learning here, and with a bit of steady work, these concepts will start to feel like second nature.
To know more in detail from our expert, watch our YouTube video:
Frequently Asked Questions
What is the mathematical representation of the First Law?
The equation is ΔE = Q - W, where ΔE is the change in energy, Q is the heat added to the system, and W is the work done by the system.
Is energy conservation the same as energy transformation?
No. Energy conservation refers to the total energy of an isolated system remaining constant, while energy transformation refers to the process of energy changing from one form (e.g., electrical) to another (e.g., mechanical).
What happens to the total energy of an isolated system?
According to the First Law, the total energy of an isolated system remains constant over time.
How do thermodynamic processes differ?
Processes are categorized by what remains constant:
-
Isobaric: Constant pressure.
-
Isochoric: Constant volume.
-
Isothermal: Constant temperature.
How is work done by a system calculated?
The work done by a system, particularly in expansion or compression, is often calculated as W = PΔV, where P is pressure and ΔV is the change in volume.
How is heat transfer ($Q$) defined?
Heat transfer occurs due to a temperature difference and can be calculated as Q = mcΔT, where m is mass, $c$ is specific heat capacity, and ΔT is the change in temperature.
What are cyclic processes?
These are processes where a system returns to its initial state, meaning the change in internal energy (ΔU) over a complete cycle is zero.
What is the Coefficient of Performance (COP)?
The COP is a measure of the efficiency of a refrigerator or heat pump, directly related to the First Law, representing the ratio of the desired cooling effect to the work input.
Why are cycles like Carnot, Otto, and Rankine important?
These cycles describe how heat is converted into work in engines and power plants. Mastering them is essential for optimizing energy efficiency in mechanical and electrical engineering.
Why is thermodynamics a priority for IIT JAM/GATE/CSIR NET?
It is a fundamental pillar of physics. These exams frequently test the ability to apply laws to various systems to determine energy balance, efficiency, and work done.
Which textbooks are recommended for IIT JAM Physics?
Standard textbooks like Halliday-Resnick-Walker and Knight are highly recommended for providing a comprehensive and in-depth understanding of thermodynamic principles.
How should one prepare for thermodynamics exam questions?
Focus on understanding the conceptual definitions first, then move to applying the ΔU = Q - W equation across different processes (isobaric, adiabatic, etc.). Practice solving problems under timed conditions.
How do practice questions and mock tests help?
They reinforce your grasp of the material, expose you to different types of problem-solving scenarios, and improve your speed and accuracy under exam pressure.
How does a power plant use the First Law?
Power plants utilize cycles (like the Rankine cycle) to convert thermal energy from fuel combustion into steam, which drives a turbine to generate electrical energy.










