Internal energy and enthalpy are fundamental concepts in thermodynamics that help in understanding the energy changes in a system. For IIT JAM, students need to grasp the definitions, units, and applications of internal energy and enthalpy to solve problems efficiently.
Syllabus – Thermodynamics and Statistical Mechanics for IIT JAM
In the world of the IIT JAM syllabus, you’ll find thermodynamic potentials—which include our stars, internal energy and enthalpy—hanging out in Unit 2.5 (and popping up again in Unit 3.3). If you want the “holy grail” of explanations, most of us at VedPrep swear by P.W. Atkins’ Physical Chemistry or C.N.R. Rao’s Modern Physics and Chemistry. They’re classic for a reason: they take these big ideas and actually make them make sense.
Internal energy and Enthalpy For IIT JAM
Let’s break these down like we’re chatting over a quick cutting chai between classes.
Internal energy (U) is basically the “bank account” of energy inside a system. It’s the sum of everything happening on a microscopic level—the kinetic energy of molecules zooming around, the potential energy from them pulling on each other, and even the energy tucked away in their bonds.
Enthalpy (H), however, is a bit more inclusive. It’s the internal energy plus the “room” the system needs to exist. We define it as
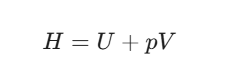
Think of it this way: if you’re inflating a balloon, you don’t just need the energy to put air inside (U); you also need the energy to push the surrounding atmosphere out of the way (pV).
Both are state functions. This is great news for you in the exam hall because it means you only care about where you started and where you ended. You don’t need to stress about the messy path the reaction took to get there.
The Key Difference: Why Does the pV Term Matter?
The main reason students get tripped up is thinking these two are the same thing. They aren’t! The big difference is that pV term. In a lab (or an exam question), if you’re doing something at constant volume, you’re looking at internal energy. As per Internal Energy and Enthalpy, if you’re doing something at constant pressure (like most chemistry experiments in an open beaker), you’re looking at enthalpy.
Worked Example: Internal Energy Change of an Ideal Gas
Let’s look at a classic problem you might see in an IIT JAM paper.
Imagine you have 2 moles of an ideal gas. You heat it up from 300 K to 400 K, but you keep it in a rigid container (constant volume). If the molar heat capacity (Cv) is 20.8 J/mol·K, what’s the change in internal energy (ΔU)?
Since the volume isn’t changing, we use the formula:
-
n = 2 moles
-
Cv = 20.8 J/mol·K
-
ΔT = 400 – 300 = 100 K
Application: Enthalpy Change in Chemical Reactions
We talk about enthalpy change (ΔH) a lot in chemistry because we usually work under atmospheric pressure. Whether it’s a combustion reaction or a phase change, ΔH tells us if the reaction is going to give off heat (exothermic) or soak it up (endothermic).
A Quick Story (The “Piston” Scenario):
Imagine a fictional experiment where you have a gas reacting inside a cylinder with a movable piston. As the gas reacts, it expands and pushes the piston up. Based on Internal Energy and Enthalpy, if you only measured the internal energy change, you’d miss the energy the gas spent “working” to lift that piston. That’s exactly why we use enthalpy—it accounts for that extra work against the outside pressure. At VedPrep, we find that visualizing these little mechanical “side quests” helps to solve problems of Internal energy and enthalpy.
Exam Strategy: Focus on Thermodynamic Potentials
A recommended study method is to practice problems involving thermodynamic potentials, which will improve understanding and problem-solving skills. This can be achieved by working through a variety of practice questions and past-year problems. VedPrep offers expert guidance and comprehensive study materials to help students prepare effectively.
When you’re staring down the IIT JAM, examiners love to test how these potentials relate to each other. Don’t just learn the formulas; understand the “why.” You’ll likely see questions on:
-
Relationships between U, H, and Helmholtz free energy (A).
-
How these change during isothermal vs. adiabatic processes.
-
Calculating ΔH from ΔU using the ideal gas law (pΔV = Δng RT).
The best way to get fast at this is just plain old practice. Flip through some past papers and try to spot where they’re trying to trick you by switching between constant pressure and constant volume.
Internal energy and enthalpy: Relationship and Significance
To wrap it up, the relationship is usually written as:
In short:
-
ΔU = Heat at constant volume (qv)
-
ΔH = Heat at constant pressure (qp)
VedPrep provides a thorough understanding of these concepts through its expert faculty and well-structured study materials. By focusing on thermodynamic potentials and practicing problems, students can build a strong foundation in thermodynamics and improve their chances of success in the IIT JAM exam.
Real-World Application: Enthalpy Change in Power Plants
Enthalpy change in power plants, where it is used to measure the energy change during various thermodynamic processes. Understanding the concept of Internal energy and enthalpy is essential for optimizing the efficiency of power plants and designing more efficient processes. The application of enthalpy change in power plants is a significant area of research and development, and it has numerous practical implications for the energy industry.
This isn’t just for passing exams. Engineers use internal energy and enthalpy to design massive steam turbines in power plants. They need to know exactly how much energy is tucked away in the steam to turn those blades and keep the lights on.
Conclusion
Mastering internal energy and enthalpy is a massive win for your IIT JAM prep. Once you get the hang of the H = U + pV relationship, a lot of the “scary” thermodynamics problems start to fall into place. Keep practicing, keep your head in the game, and you’ll find that these concepts become your best friends on exam day.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
How is enthalpy (H) defined?
Enthalpy is a measure of the total heat content of a system. It is mathematically defined as the sum of internal energy (U) and the product of pressure (p) and volume (V): H = U + pV.
What are the SI units for internal energy and enthalpy?
Both internal energy and enthalpy are forms of energy, so their standard SI unit is the Joule (J).
Are internal energy and enthalpy state functions?
Yes, both internal energy and enthalpy are state functions. This means their values depend solely on the current state of the system (defined by variables like pressure, volume, and temperature) and are independent of the path taken to reach that state.
What does "thermodynamic potential" mean in the context of IIT JAM?
Thermodynamic potentials, such as internal energy, enthalpy, and Helmholtz free energy, are scalar quantities used to represent the thermodynamic state of a system and predict the energy changes during processes.
What is the key difference between internal energy and enthalpy?
The fundamental difference is the inclusion of the pV term in enthalpy, which accounts for the energy associated with the system's pressure-volume work.
Can internal energy and enthalpy be used interchangeably?
No. While both are thermodynamic properties, they are not interchangeable. Internal energy represents the system's inherent energy, whereas enthalpy includes the energy required to "make room" for the system in its environment (the pV work).
Why is the distinction between state and path functions important?
Understanding that internal energy and enthalpy are state functions allows students to calculate changes based only on initial and final states, simplifying complex thermodynamic calculations significantly.
How does enthalpy change relate to heat transfer?
At constant pressure, the change in enthalpy (ΔH) is exactly equal to the heat transferred (qp) to or from the system.
How do you calculate enthalpy change during a phase transition?
You can use the relationship ΔH = ΔU + pΔV. By determining the change in internal energy and the change in volume during the transition (e.g., melting ice to water), you can calculate the total enthalpy change.
What role does calorimetry play in measuring enthalpy?
Calorimetry is the experimental technique used to measure the heat flow associated with chemical reactions or physical changes, which directly allows for the determination of ΔH.
Where do thermodynamic potentials fall in the IIT JAM syllabus?
Thermodynamic potentials, including internal energy and enthalpy, are typically covered under Unit 2.5 of the Thermodynamics and Statistical Mechanics section of the IIT JAM syllabus.
Which textbooks are recommended for studying this topic?
Students should refer to Physical Chemistry by P.W. Atkins (8th ed.) and Modern Physics and Chemistry by C.N.R. Rao (3rd ed.) for comprehensive coverage.
What subtopics of thermodynamic potentials are frequently tested in IIT JAM?
Commonly tested areas include definitions and fundamental equations, relationships between potentials (U, H, A, G), and their specific applications to thermodynamic processes.
How can practicing problems improve my score in thermodynamics?
Solving varied problems, including past-year questions, helps in mastering the application of formulas and ensures students can quickly identify which potential is relevant to a specific thermodynamic system.










