Physical properties of liquids (Vapour pressure) For IIT JAM, such as vapour pressure, surface tension, and viscosity play a critical role in understanding the behavior and applications of liquids in various fields, making it a vital topic for IIT JAM aspirants.
Physical properties of liquids (Vapour pressure) For IIT JAM
If you’ve ever left a bottle of hand sanitizer open and noticed it “disappeared” faster than a bottle of water, you’ve already started studying the physical properties of liquids. For anyone eyeing a seat in a top IIT through the JAM 2027 cycle, mastering concepts like Physical properties of liquids, surface tension, and viscosity isn’t just about ticking off a syllabus box—it’s about understanding why liquids behave the way they do in a lab and in the real world.
The process of getting comfortable with these ideas starts with looking at pure substances by covering Physical properties of liquids. Whether you’re prepping for IIT JAM, CSIR NET, or GATE, you really need to wrap your head around how vapour pressure works. The IIT JAM syllabus explicitly lists the “Physical and Chemical Properties of Pure Substances” as a core area.
Simply put, the Physical properties of liquids is the pressure exerted by a vapour when it’s in a happy equilibrium with its liquid or solid form at a specific temperature. If you want to dive deeper into the math and theory, P. W. Atkins or I. H. Greenwood’s Physical Chemistry are the gold standards. At VedPrep, we often suggest these as go-to resources because they break down the “why” behind the curves and equations you’ll see on exam day.
Importance of Physical properties of liquids (Vapour pressure) For IIT JAM: Applications and Concepts
Physical properties of liquids isn’t just a number in a table; it’s the secret sauce behind many lab techniques. Take distillation, for example. If you’re trying to separate two liquids, you’re basically playing a game of “who turns into a gas first?” The liquid with the higher vapour pressure (the more volatile one) leaves the flask sooner.
In solvent extraction, vapour pressure helps you pick the right liquid for the job. If a solvent is too volatile, it might evaporate before it finishes its work; too stable, and you’ll struggle to get it out of your final product.
This concept also helps us predict boiling points. A liquid boils when its internal Physical properties of liquids match the pressure pushing down on it from the atmosphere. This is why water boils faster (and at a lower temperature) in the mountains than it does at sea level. If you’re looking into industrial heat transfer or fluid flow, these basics are non-negotiable.
Even when things get messy with non-ideal solutions—where molecules don’t always play nice together—analysing vapour pressure data gives us a window into the thermodynamics of the mixture.
Physical properties of liquids (Vapour pressure) For IIT JAM
To really nail those JAM questions, you’ve got to move past the definition. You need to see how the Physical properties of liquids transform as per environment. At VedPrep, we recommend a two-step approach:
-
Get the basics down cold (what is equilibrium, really?).
-
Hammer out practice problems, especially those involving the Clausius-Clapeyron equation.
Understanding how intermolecular forces—like those pesky hydrogen bonds—hold molecules back from escaping into the air is key. The stronger the “hug” between molecules, the lower the vapour pressure. By following a structured study plan and utilizing resources like VedPrep, students can develop a deep understanding of vapour pressure and its applications in Vapour pressure For IIT JAM, ultimately achieving success in IIT JAM and other competitive exams.
Physical properties of liquids (Vapour pressure) For IIT JAM: Common Misconceptions
One big trap students fall into is treating vapour pressure and atmospheric pressure like they’re the same thing. They aren’t. Atmospheric pressure is the weight of the air above you; vapour pressure is the “escape push” of the liquid itself.
Another myth is that vapour pressure is a fixed constant. It’s actually very moody—it changes as soon as you change the temperature. Think about a cup of hot chai versus a glass of ice water. The hot chai has much more “steam” (vapour) coming off it because the higher temperature gives molecules the energy to break free.
Example: Compare water and ethanol. Water is like a group of friends holding hands tightly (hydrogen bonding), so it has a lower vapour pressure. Ethanol is a bit more relaxed, with weaker forces, so its molecules jump into the gas phase much more easily.
Worked Example: Calculating Vapour Pressure using Raoult’s Law
Raoult’s Law is your best friend for ideal solutions. It tells us that the partial pressure of a component is just its “pure” pressure multiplied by how much of it is in the mix (mole fraction).
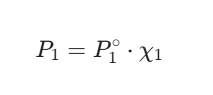
-
Let’s imagine a fictional scenario: You’ve got a solution that is 20% ethanol and 80% water (by mole) at 25°C.
-
Pure water pressure (P°water): 23.76 mmHg
-
Pure ethanol pressure (P°ethanol): 58.9 mmHg
To find the total vapour pressure, you’d calculate:
Ptotal = (0.80 × 23.76) + (0.20 × 58.9)Ptotal = 19.01 + 11.78 = 30.79 mmHg -
Surface Tension and Viscosity: Related Properties of Liquids
While vapour pressure is about “escaping,” surface tension is about “holding on.” It’s the energy needed to stretch the surface of a liquid. Imagine a water strider walking on a pond—that’s surface tension in action. It happens because the molecules on top don’t have neighbours above them to pull on, so they pull tighter on the ones beside and below them.
Viscosity is just a fancy word for “thickness” or resistance to flow. Think of the difference between pouring water and pouring honey. Honey has high viscosity; it’s sticky and slow. Both of these properties depend heavily on intermolecular forces and temperature. If you heat up honey, it gets “thinner” and flows faster because the heat helps the molecules slide past each other more easily.
Real-World Applications : Examples and Case Studies
Engineers use vapour pressure to design the massive distillation columns that give us gasoline and medicine. Without accurate vapour pressure data, these towers wouldn’t work, and we’d be stuck with impure products.
Even your laundry detergent depends on this. Companies have to balance the “volatility” of the scents and cleaning agents. If the vapour pressure of the fragrance is too high, your clothes stop smelling fresh five minutes after they dry. If it’s too low, you won’t smell anything at all.
Lastly, meteorologists use these same principles to predict the weather. Humidity is essentially just the vapour pressure of water in our atmosphere. When that pressure changes, it’s the difference between a sunny day and a thunderstorm.
Final Thoughts
Mastering the Physical properties of liquids (Vapour pressure) is more than just a syllabus requirement; it is a foundational step toward excelling in the physical chemistry sections of IIT JAM 2027. By understanding how molecular interactions and temperature dictate macro-level behaviors like evaporation and boiling, aspirants can transition from rote memorization to analytical problem-solving. Whether you are calculating deviations using Raoult’s Law or predicting distillation efficiency, a clear conceptual grasp of these properties will be your greatest asset. As you continue your preparation, focus on the interconnectedness of these liquid states to build the academic rigor necessary for a top-tier rank.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
Is vapour pressure the same as atmospheric pressure?
No. Vapour pressure is an intrinsic property of a substance based on its temperature and molecular makeup, while atmospheric pressure is the external pressure exerted by the weight of the air above us.
What is the relationship between temperature and vapour pressure?
They are directly proportional. As temperature increases, the kinetic energy of molecules increases, allowing more of them to escape into the gas phase, which raises the vapour pressure.
How do intermolecular forces affect vapour pressure?
There is an inverse relationship. Stronger intermolecular forces (like hydrogen bonding in water) hold molecules together more tightly, resulting in a lower vapour pressure compared to liquids with weak forces.
What does "volatile" mean in the context of vapour pressure?
A volatile liquid is one that has a high vapour pressure at room temperature, meaning it evaporates easily (e.g., acetone or ethanol).
When does a solution follow Raoult’s Law?
A solution follows Raoult’s Law when it is an ideal solution, meaning the interactions between different molecules (A-B) are similar to the interactions between like molecules (A-A and B-B).
What is the Clausius-Clapeyron equation used for?
It is used to calculate the vapour pressure of a substance at different temperatures or to determine the enthalpy of vaporization (ΔHvap) by measuring how vapour pressure changes with temperature.
What is "Relative Lowering of Vapour Pressure"?
It is a colligative property where the addition of a non-volatile solute reduces the vapour pressure of the solvent. The ratio of this decrease to the pure solvent's pressure equals the mole fraction of the solute.
Do all liquids follow Raoult's Law?
No. Non-ideal solutions show "positive" or "negative" deviations depending on whether the intermolecular attractions between the solute and solvent are weaker or stronger than in the pure components.
How is boiling point defined in terms of vapour pressure?
The boiling point is the temperature at which the vapour pressure of the liquid becomes equal to the external atmospheric pressure.
Why do liquids boil at lower temperatures at high altitudes?
At high altitudes, atmospheric pressure is lower. Since a liquid only needs to reach a lower vapour pressure to match the surrounding pressure, it boils sooner.
What is the "Normal Boiling Point"?
The normal boiling point is the temperature at which a liquid's vapour pressure equals exactly 1 atm (760 mmHg).
What happens to vapour pressure at the critical point?
At the critical point, the distinction between the liquid and gas phases disappears, and the vapour pressure reaches its maximum value where the two phases coexist.
How are surface tension and vapour pressure related?
Both are governed by intermolecular forces. Generally, a liquid with high surface tension (stronger forces) will have a lower vapour pressure.
Does viscosity change with vapour pressure?
Not directly, but they share a common cause: temperature. Increasing temperature decreases viscosity (easier flow) and increases vapour pressure (easier evaporation).














