Planck’s quantum theory propose that light energy is released or taken up in separate units termed quanta, instead of as a steady wave. This core notion established the foundation for present-day quantum mechanics by presenting quantized energy, which suggests a photon’s energy is directly proportional to its frequency.
Origin of Planck’s quantum theory in physics
Nineteenth-century physics couldn’t account for the way heated objects radiate energy at particular strengths across various light spans. Maxwell’s electromagnetic wave concepts at the close of the 1800s proved insufficient for explaining both blackbody emission and the emission of electrons when light strikes a material. Max Planck resolved this problem by proposing that energy exchanges occur in distinct bundles. This advance signified the termination of the predictable, machine-like depiction of the universe and introduced the domain of physics related to the atomic level. Applicants need to concentrate on subjects such as Planck’s quantum theory by reviewing the I IIT JAM Chemistry Syllabus to achieve a top mark.
Scientists noted that conventional mechanics failed when applied to tiny particles. Research showed that the heat retention of solid materials varied with temperature in ways Newtonian ideas could not explain. Max Planck introduced his hypothesis to align experimental results with current models. His work shifted the focus of research away from continuous wave motion toward discrete particle attributes.
Fundamental postulates of quantization of energy
Planck’s quantum theory relies on several core pillars that define how matter interacts with radiation. The primary postulate states that hot bodies emit radiant energy in a discontinuous manner. This energy travels in small units known as quanta or photons. A body cannot emit or absorb a fraction of a quantum; it must be a whole number multiple of the base energy unit.
This restriction is known as the quantization of energy. It means a system can only exist in specific energy states. For example, a body might emit hν or 2hν but never 1.5hν. This discovery changed the understanding of atomic and molecular structure by proving that energy levels are restricted. You can observe this principle in modern electronics where electrons transition between fixed energy gaps.
This constraint is termed the quantization of energy. It implies that a system is confined to discrete energy levels. For instance, a body might emit hν or 2hν but never 1.5hν. This finding altered the comprehension of atomic and molecular makeup by demonstrating that energy steps are limited. This concept is visible in contemporary electronics where electrons jump across defined energetic separations.
Mathematical expression of Planck’s law
Planck’s quantum theory offers a measurable account of blackbody spectral emission. The energy E of an individual quantum connects to the radiation’s frequency ν via a multiplicative factor. This association is central to quantum science and facilitates energy computation across the electromagnetic range.
| Component | Mathematical Representation |
|---|---|
| Energy Frequency Relation | E = hν |
| Wavelength Relation | E = hc/λ |
| Planck’s Constant (h) | 6.626 ×10-34 J s |
| Speed of Light (c) | 3.0 ×108 m/s |
| Boltzmann Constant (k) | 1.381 × 10-23 J/K |
| Blackbody Spectral Radiance | 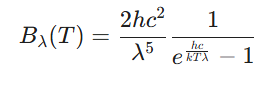 |
Role of blackbody radiation in quantum mechanics
An ideal blackbody both takes in all arriving radiation and radiates energy at the highest rate for a given temperature. Planck’s quantum theory accurately described the strength of this emission across every frequency. Earlier theories forecast an unending rise in intensity at shorter wavelengths, an issue termed the ultraviolet catastrophe. Planck’s formula fixed this by demonstrating that higher frequency energy packets demand greater input energy.
Practical applications of Planck’s quantum theory
Modern technology relies on the principles like Planck’s quantum theory. Lasers stand as a chief illustration where illumination is output in separate bundles. Within optical transmission, laser rays convey vast amounts of data rapidly due to light’s quantum characteristic. Every photon bears a distinct energy profile enabling exact signal management across fiber optic systems.
Aside from just conversations, this concept spurred both transistors and diodes into existence. Such elements underpin every computer chip and mobile phone. The advance of quantum electronics originates from managing subatomic energy stages within semiconducting materials. Lacking comprehension of distinct energy parcels, building today’s microprocessors would be infeasible.
Connection to atomic and molecular structure
The quantization of energy fundamentally shapes how researchers depict electron configurations within atoms. Planck’s quantum theory posits that electrons reside in defined paths possessing set energy magnitudes. Should an electron transition between these states, it either takes in or releases a photon matching a precise frequency. This characteristic accounts for the distinct spectral signatures noted for various chemical elements.
This notion is vital for learners going over IIIT JAM Chemistry PYQs. Entry tests frequently demand determining the energy of shifts or pinpointing the wavelength of light discharged. Grasping that energy lacks a steady flow aids in tackling issues about ionization potential and electron layers. This framework offers the guidelines that manage the endurance and response of every chemical entity.
Limitations of the original quantum hypothesis
Planck put forth quantization as a numerical remedy for blackbody emissions rather than a full physical model. He didn’t at first detail the underlying physical reasons for energy needing to be discrete. His initial concept centered on the radiation field, leaving out the quantization of momentum or rotational momentum. Furthermore, it didn’t capture the matter’s wave-particle nature.
Initial quantum ideas viewed light as separate elements while overlooking its wave characteristics in specific scenarios. A more advanced structure, featuring Schrödinger’s equation and matrix mechanics, eventually became necessary to cover these omissions. The initial concept failed to forecast intricate atomic arrangements or account for entanglement and superposition. These theoretical problems persisted until complete quantum mechanics, like Planck’s quantum theory, emerged.
Critical perspective on the classical vs quantum transition
A widespread misunderstanding suggests quantum mechanics instantly superseded classical physics across the board. Actually, the classical mechanics model retains its precision for larger-scale objects. Planck’s quantum theory become essential when examining events at the atomic level. The transition between these two worlds occurs when the action of a system is comparable to Planck’s constant.
Applying established rules for tiny particles results in considerable inaccuracies. For example, the conventional model fails to account for why electrons maintain their orbits instead of spiraling inward toward the core. Although traditional physics implies energy can assume any level, quantum mechanics’ nature of separated states stops atomic disintegration. You must apply quantum principles when the discrete nature of energy impacts the total behavior of the system. You must consider knowledge on Planck’s quantum theory to understand nature of energy.
Impact on the photoelectric effect
Einstein built upon Planck’s quantum theory to account for the photoelectric phenomenon. He suggested that light is made up of discrete units termed photons. When these photons hit a metal face, they pass their energy to electrons. Should a photon’s energy surpass the electron’s attachment energy, the electron is released.
This empirical verification demonstrated light’s particle nature. Conventional wave models suggested that boosting light power would eventually knock out electrons irrespective of their frequency. Tests indicated that if the frequency remains below a certain threshold, virtually no electrons emerge, regardless of how intense the illumination is. This result substantiated Planck’s concept that energy is tied to frequency, rather than solely to the wave’s magnitude.
Influence on future scientific advancements
The finding of quantization signaled a critical moment in scientific progress. It facilitated the creation of the photon idea and paved the way for Bohr’s atomic structure. Contemporary areas such as quantum computation and the study of solids trace their roots back to Planck’s first supposition. These leaps forward keep influencing your engagement with digital tools and diagnostic visualization.
Inquiries within quantum mechanics now tackle intricate problems such as measurement and entanglement within Planck’s quantum theory. Researchers employ the bedrock of Planck’s findings to investigate the characteristics of antimatter and infinitesimal particles like quarks and leptons. The transition from a deterministic cosmos to one defined by probability stands as the foremost alteration in humanity’s perception of the physical realm.
Final Thoughts
Planck’s quantum theory stands as the essential model for grasping the shift from classical mechanics to the granular nature of the atomic scale. By demonstrating that energy isn’t smooth but comes in distinct bundles known as quanta, this theory unraveled the puzzle of luminous body emission and paved the way for contemporary innovations such as lasers and transistors. At VedPrep, we offer tailored materials and professional support to assist you in conquering these intricate ideas for your rigorous assessments. Grasping the fundamental principles of energy quantization is a crucial prerequisite for any pupil hoping to succeed in advanced academic entry examinations.
Learn more from our expert Physics professionals, watch our Youtube video:
Frequently Asked Questions (FAQs)
What is Planck’s quantum theory?
Planck’s quantum theory states that energy is not continuous but exists in small, discrete packets called quanta. Max Planck proposed this theory to explain blackbody radiation, proving that energy emissions occur in finite units. This principle shifted physics from classical Newtonian mechanics to the study of atomic and subatomic levels.
Who proposed Planck’s quantum theory?
German physicist Max Planck introduced this theory in the early 20th century. His work addressed the failures of classical physics, particularly regarding how hot bodies emit radiation. This discovery earned him a central place in the history of modern science and quantum mechanics.
What is a quantum in Planck’s theory?
A quantum is the smallest possible unit or packet of energy that a body can emit or absorb. In the context of light, these packets are often called photons. The energy of each quantum remains directly proportional to the frequency of the radiation.
How does Planck’s theory define energy emission?
Planck’s theory defines energy emission as a discontinuous process occurring in small, separate units. A body transmits thermal radiation in specific amounts rather than a steady, unbroken stream. This concept of discrete steps is a fundamental shift from classical wave theories.
What is the relationship between energy and frequency?
The energy of a quantum is directly proportional to the frequency of the emitted radiation. As the frequency increases, the energy per photon also increases. This relationship is expressed by the formula E = hν, where h is Planck’s constant.
What is the value of Planck’s constant?
Planck’s constant, denoted as $h$, has a standard value of 6.626 × 10-34 J s. This fundamental constant determines the size of the energy packets in quantum mechanics. It appears in almost every equation involving the quantization of energy at the atomic scale.
How do you calculate energy using wavelength?
You calculate energy from wavelength using the equation E = hc/λ. In this formula, $h$ is Planck’s constant, c is the speed of light, and λ is the wavelength. This allows you to determine energy when frequency is not directly provided.
What is the speed of light in a vacuum?
The speed of light in a vacuum is approximately 3.0 ×108 m/s. This constant value is used in Planck’s equations to relate frequency and wavelength. It remains consistent regardless of the frequency of the electromagnetic radiation.
How is quantization applied to energy absorption?
A body absorbs energy only in whole number multiples of hν, such as 1hν, 2hν, or 3hν. It cannot absorb partial quanta. This ensures that energy transitions in atoms and molecules remain discrete and predictable.
Why did classical physics fail to explain blackbody radiation?
Classical physics predicted that hot bodies would emit infinite energy at shorter wavelengths, which did not happen in experiments. This was known as the ultraviolet catastrophe. Planck’s quantum theory corrected this by proposing that energy is restricted to discrete packets.
Does Planck’s theory address the dual nature of light?
The original theory by Planck focused on energy packets but did not fully incorporate wave properties. Later developments in quantum mechanics merged these ideas into wave particle duality. Planck’s work provided the necessary starting point for this broader understanding.
What are the main limitations of Planck’s original theory?
Planck’s initial theory was empirical and lacked a full mathematical framework for all physical quantities. It did not explain the quantization of momentum or angular momentum. Furthermore, it did not provide a statistical framework for matter like electrons.
How does Planck’s theory explain the photoelectric effect?
Albert Einstein used Planck’s idea of quanta to show that light particles, or photons, eject electrons from metal surfaces. Each photon must have enough energy, determined by its frequency, to release an electron. This provided critical experimental proof for Planck's hypothesis.
What are the implications for IIT JAM Chemistry PYQs?
Competitive exams like IIT JAM frequently test your ability to calculate photon energy and understand atomic transitions. Questions often involve the relationship between frequency, wavelength, and the energy levels of electrons. Mastering these calculations is essential for scoring well in physical chemistry sections.













