The Uncertainty Principle, a core concept in quantum mechanics, dictates that the simultaneous exact measurement of a particle’s location and impetus is impossible. This connection suggests that gaining precision in determining position inevitably reduces the precision of the momentum measurement, setting a fundamental constraint within the physical world.
Physical Basis of the Heisenberg Uncertainty Principle formula
The Uncertainty Principle stems from the wave-particle nature of entities such as electrons. In classical physics, you can determine exactly where an object is and how fast it moves. Quantum mechanics replaces these definite paths with wave functions, where the mathematics of continuous spectra replaces discrete sets. A wave function that defines a specific position must consist of many different wavelengths. Since momentum relates directly to wavelength through the de Broglie formula, a defined position results in a wide spread of possible momenta. The Heisenberg Uncertainty Principle formula mathematically shows this trade-off as follows:
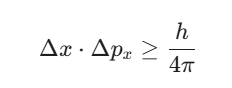
Within this formula, Δx denotes the imprecision in location and Δpx signifies the uncertainty in the product of mass and velocity. The constant h is Planck’s constant. You can also write this using ![]() , where
, where ![]() , leading to the form
, leading to the form ![]() . This equation shows that the product of these uncertainties has a lower limit. You cannot reduce one uncertainty to zero without making the other infinite.
. This equation shows that the product of these uncertainties has a lower limit. You cannot reduce one uncertainty to zero without making the other infinite.
Role in the IIT JAM Chemistry Syllabus
The IIT JAM Chemistry Syllabus necessitates a thorough grasp of how quantum limitations govern the arrangement of atoms. The principle of uncertainty clarifies why electrons avoid spiraling inwards toward the nucleus. Should an electron be restricted to a minute nuclear region, its positional uncertainty would be exceptionally small. Per the Heisenberg Uncertainty Principle formula, its momentum uncertainty would consequently become vast. This substantial momentum would furnish the electron with ample energy to break free from the nuclear attraction.
Calculations involving this principle often appear in the IIT JAM Chemistry PYQs. You must be able to calculate minimum uncertainties for electrons in atoms versus macroscopic objects. For an electron in a hydrogen atom, the uncertainty in velocity is often comparable to the velocity itself. For a one-gram item, the uncertainty is so minuscule it stays unnoticed. This difference clarifies why classical mechanics applies to bigger objects, while quantum mechanics is essential for particles at the subatomic level.
Mathematical Treatment and Energy Time Relationship
A different version of the uncertainty principle deals with energy and time. This formulation is crucial for grasping the breadth of spectral lines as described in the IIT JAM Chemistry Syllabus. If a quantum state exists for a finite time Δt, its energy ΔE cannot be perfectly defined. The relationship follows a similar mathematical structure:
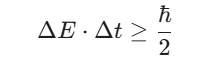
This energy spread is known as natural broadening. A state with a very short lifetime will have a large uncertainty in its energy. When you analyze IIT JAM Chemistry PYQs, you might encounter problems asking for the lifetime of an excited state based on the width of a spectral line. This application proves that the Uncertainty Principle is not just a measurement difficulty but an intrinsic property of quantum systems.
Comparison of Uncertainty Applications
The following table outlines how the Uncertainty Principle applies to different physical scenarios and the resulting implications for your exam preparation.
| Scenario | Physical Constraint | Resulting Observation |
|---|---|---|
| Electron in an Atom | Small Δx (approx. 10-10 m) | High velocity uncertainty (106 m/s) |
| Macroscopic Ball | Large Δx (approx. 10-6 m) | Negligible velocity uncertainty (10-25 m/s) |
| Sit Diffraction | Narrow slit width (b) | Increased angular spread of particles |
| Atomic Transitions | Finite lifetime (τ) | Natural broadening of spectral lines |
Information Gain through Experimental Evidence
Heisenberg originally illustrated this concept using a hypothetical gamma ray microscope. To see an electron, you must bounce a photon off it. A short wavelength photon provides a better position measurement. However, short wavelength photons carry high momentum. When the photon hits the electron, it transfers a random amount of momentum through the Compton effect.
This engagement alters the electron’s movement without a set pattern. Employing a photon with greater wavelength lessens the momentum shift, yet it makes the electron’s location fuzzier. You confront a decision between ascertaining the particle’s whereabouts or its trajectory. This conceptual exercise demonstrates that the process of measurement inherently perturbs the quantum arrangement.
Limitations and Common Misconceptions in Quantum Theory
A frequent mistake is thinking the Uncertainty Principle stems from inadequate apparatus. This isn’t right. Even utilizing flawless equipment, the fuzziness persists because it’s a mathematical outcome of wave mechanics. Another mistake is applying the principle to variables that are compatible. As an illustration, accurately determining both the x-coordinate and the y-momentum is feasible since they are not mutually exclusive measurements.
In the context of the IIT JAM Chemistry Syllabus, you must distinguish between the Uncertainty Principle and the observer effect. While the gamma ray microscope involves an observer effect, the fundamental uncertainty exists even without a physical probe. This characteristic belongs to the wave function’s nature. Grasping this difference is crucial for achieving good marks on conceptual queries in IIT JAM Chemistry PYQs.
Practical Problem Solving for IIT JAM
When solving numerical problems, always check your units. Planck’s constant is 6.626 × 10-34 J·s. For an electron, use the mass 9.1 × 10-31 kg. Most exam problems ask for the “minimum uncertainty,” which means you should treat the inequality as an equality.
When a problem gives a velocity percentage, determine the absolute uncertainty in velocity, Δv, initially. Subsequently, apply Δp = mΔv within the Heisenberg Uncertainty Principle equation. Following this organized method guarantees precision when dealing with the quantum mechanics part of the IIT JAM Chemistry syllabus. Familiarize yourself with the magnitude of these quantities by working through previous year questions from IIT JAM Chemistry.
Quantum Operators and Non-Commutation
The Uncertainty Principle is closely tied to the axioms of quantum mechanics. Within the mathematical framework of the IIT JAM Chemistry Syllabus, physical quantities are signified by operators. Two observables have a simultaneous measurement limit if their operators do not commute, meaning they have different state functions.
For position ![]() and momentum
and momentum ![]() , the commutator results in a non-zero value. Because this value is non-zero, the two properties are incompatible. If you find the commutator of two operators to be zero, you can measure those properties at the same time without any uncertainty limit. This algebraic foundation is a frequent topic in advanced IIT JAM Chemistry PYQs and forms the basis for understanding more complex quantum systems.
, the commutator results in a non-zero value. Because this value is non-zero, the two properties are incompatible. If you find the commutator of two operators to be zero, you can measure those properties at the same time without any uncertainty limit. This algebraic foundation is a frequent topic in advanced IIT JAM Chemistry PYQs and forms the basis for understanding more complex quantum systems.
Conclusion
The Heisenberg Uncertainty Principle is a core constraint of reality stemming from the wave-like and particle-like nature of stuff. It dictates that as you better determine a particle’s location, the accuracy with which its momentum can be ascertained decreases, and the opposite is also true. Although these influences are minor for large objects, they are crucial at the atomic level and vital for grasping how elementary particles act.
Grasping these quantum mechanics principles and their mathematical applications is vital for students pursuing the IIT JAM Chemistry curriculum. VedPrep offers thorough materials and professional support to assist you in tackling these intricate subjects and succeeding in your admission test readiness. By viewing the Uncertainty Principle as an inherent feature of quantum entities instead of a flaw in measurement, you achieve a more profound, precise outlook on the subatomic realm.
To learn more from our experts, watch our Youtube video:
Frequently Asked Questions (FAQs)
What is the Uncertainty Principle?
The Uncertainty Principle is a fundamental law in quantum mechanics stating you cannot simultaneously measure the exact position and momentum of a particle. This limit is a property of nature rather than a result of technological flaws. It emerges because particles like electrons exhibit wave like behaviors.
How does the Heisenberg Uncertainty Principle formula work?
The Heisenberg Uncertainty Principle formula states that the product of uncertainty in position and momentum must be at least h divided by 4 pi. The mathematical expression is Delta x times Delta p is greater than or equal to h cross divided by two. This relationship dictates that as one measurement becomes more precise, the other becomes less certain.
What causes the Uncertainty Principle?
This principle arises from wave particle duality. Because particles behave like waves, their position and momentum are described by wavefunctions. A wavefunction representing a precise position requires many different wavelengths, which leads to a large uncertainty in momentum. It is an inherent limitation of quantum systems.
Why is the Uncertainty Principle important in chemistry?
The principle is a core part of the IIT JAM Chemistry Syllabus because it explains atomic structure. It prevents electrons from collapsing into the nucleus. If an electron were restricted to a tiny nuclear volume, its momentum would become high enough to escape the nucleus. This maintains the stability of atoms.
How does the principle relate to the de Broglie postulate?
The de Broglie formula connects momentum to wavelength, while the uncertainty principle limits the simultaneous knowledge of position and that wavelength. Heisenberg used the de Broglie postulate to derive the limits of measurement for electrons passing through a slit. Both concepts are essential for understanding wave mechanics.
How do you calculate uncertainty for an electron?
To find the uncertainty, you identify the known precision of either position or velocity. You multiply the mass of the electron by the velocity uncertainty to get momentum uncertainty. Then use the Heisenberg Uncertainty Principle formula to solve for the remaining unknown variable. Ensure all units are in the SI system.
How does slit width affect particle uncertainty?
When a particle passes through a slit of width b, its position uncertainty is roughly b. Reducing the slit width makes the position more certain but increases the diffraction or spread of the particle momentum. This demonstrates the reciprocal relationship between the two measurements.
What is the energy time uncertainty relation?
This relation states that the uncertainty in energy multiplied by the time duration of a measurement is at least h cross divided by two. It implies that states with a short lifetime have a natural spread in their energy values. You see this effect in the broadening of spectral lines.
Does the Uncertainty Principle apply to macroscopic objects?
The principle applies to all objects but the effects are negligible for large masses. A one gram object has such a small uncertainty that it cannot be measured by current instruments. The value of Planck's constant is too small to impact everyday measurements of large bodies.
Why is my uncertainty calculation different from the textbook?
Check if you used h or h cross in your formula. The standard Heisenberg Uncertainty Principle formula uses h divided by 4 pi, which equals h cross divided by two. Using the wrong constant will result in a factor of two error in your final answer.
Can technology eventually overcome the Uncertainty Principle?
No, the principle is a fundamental law of nature rather than a limitation of equipment. It is a consequence of the mathematical nature of wavefunctions. Even with theoretically perfect measuring devices, the uncertainty would remain unchanged.
What happens if position uncertainty is zero?
If the uncertainty in position is zero, the uncertainty in momentum must be infinite to satisfy the Heisenberg Uncertainty Principle formula. This means the particle could have any momentum value. In physical reality, you cannot achieve zero uncertainty for either variable.
Is the Uncertainty Principle the same as the observer effect?
They are different concepts. The observer effect refers to changes that a measurement makes to a system, like a photon hitting an electron. The Uncertainty Principle is a more fundamental property involving the wave nature of the particle itself. Heisenberg's microscope example illustrates both.
What are incompatible observables?
Incompatible observables are physical properties that cannot be known simultaneously. Position and momentum are incompatible because they have different state functions. In the mathematical treatment of quantum mechanics, their operators do not commute.
How does the principle affect the ground state energy?
The principle requires that a particle confined in a potential well, like an atom, has a minimum non zero energy. This is called zero point energy. If the energy were zero, the momentum would be exactly known, violating the uncertainty principle.













