Viscosity For IIT JAM is a fundamental concept in physics that deals with the measure of a fluid’s resistance to flow. It’s crucial for students preparing for CSIR NET, IIT JAM, CUET PG, and GATE to understand fluid dynamics and its applications, as it’s a key topic in the exam syllabus.
Viscosity For IIT JAM: Definition and Significance
Simply put, viscosity is how much a fluid fights back when you try to make it move. It’s internal friction. In your syllabus, you’ll see it represented by the Greek letter μ (mu), measured in Pascal-seconds (Pa·s) or Poise (P).
Why does it matter for the JAM? Because it tells us how molecules behave. A fluid’s “thickness” depends on its molecular structure and density. Take honey versus water: honey is a nightmare to pour because its complex molecules are essentially holding hands and refusing to let go, creating high flow resistance. In the lab, you’ll find that understanding this is key to grasping how gases behave, which is a massive chunk of the IIT JAM Chemistry syllabus.
In fluid dynamics, viscosity helps us figure out the Reynolds number. This is just a fancy way of asking, “Is this flow smooth (laminar) or a hot mess (turbulent)?” High viscosity usually keeps things chill and laminar, while low viscosity lets things get chaotic and turbulent.
Viscosity For IIT JAM: Real-World Applications
Viscosity is everywhere. Imagine engineers trying to pump oil through a pipeline that’s hundreds of miles long. If they don’t get the viscosity math right, they won’t know how much power the pump needs, and the whole system could fail.
It’s also why your AC works. The refrigerants inside have specific viscosities that change how heat moves through the coils. If the flow resistance is off, your room stays hot, and the machine works double time.
Even in your own body, viscosity is a big deal. Blood flow resistance is a huge factor in cardiovascular health. If blood gets too “thick,” your heart has to pump harder, which changes your blood pressure. Researchers use rheology (the study of flow) to design better treatments for heart disease. At VedPrep, we love these kinds of examples because they show that the math you’re doing today actually saves lives tomorrow.
Misconceptions About Viscosity
Let’s clear the air on a few things that trip up even the best students:
-
Density ≠ Viscosity: Just because something is heavy doesn’t mean it’s viscous. Mercury is incredibly dense, but it flows much faster than room-temperature pancake syrup. They’re different physical properties—don’t let the exam questions trick you into thinking they’re the same.
-
The Temperature Trap: This is a classic JAM question. For liquids, if you turn up the heat, flow resistance goes down (think of cold honey vs. microwaved honey). But for gases? It’s the opposite! Heating a gas makes the molecules zip around and collide more, which actually increases flow resistance.
-
Internal Friction: While viscosity is a measure of resistance, it’s specifically about resistance to “shear stress.” Don’t get bogged down in thinking it’s just “thickness.”
The relationship between fluid dynamics and temperature is often misunderstood. It is commonly assumed that fluid dynamics is directly related to a fluid’s temperature. In reality, viscosity typically decreases with increasing temperature for liquids, while it increases with temperature for gases. This complex behavior highlights that viscosity is not simply a function of temperature but depends on the specific properties of the fluid.
Exam Strategy: Viscosity For IIT JAM
To crush the JAM, you need to move past just memorizing the definition. You need to get comfortable with the math.
Practice problems are your best friend here. At VedPrep , we suggest focusing on:
-
Units: Know how to convert between Poise and Pa·s in your sleep.
-
Non-Newtonian Fluids: Think about oobleck or ketchup—things that don’t follow the normal rules.
-
Pipe Flow: This is where the marks are. Learn how fluids move through channels and what happens when the diameter changes.
Reviewing the applications of viscosity in real-world scenarios is vital for IIT JAM preparation. Fluid dynamics plays a significant role in various fields, including engineering, materials science, and biomedical research. For expert guidance, VedPrep offers comprehensive resources and online coaching to help students prepare for IIT JAM and other competitive exams.
Important Subtopics in Viscosity For IIT JAM
If you’re making a checklist, put Poiseuille’s Law and the Hagen-Poiseuille equation at the top
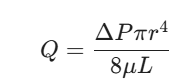
This formula looks scary, but it’s just explaining how the flow rate (Q) depends on the pressure drop, the pipe’s radius (r), and—you guessed it—viscosity (μ).
You should also get a handle on the Reynolds number (Re) and the Nusselt number. These aren’t just random names; they are the tools you’ll use to predict if a fluid will behave itself in a pipe or start swirling around like a whirlpool.
By following a structured study plan and practicing with sample questions, students can build a strong foundation in fluid dynamics and achieve success in IIT JAM and other competitive exams. Effective preparation will enable students to approach Viscosity for IIT JAM with confidence.
Lab Applications of Viscosity
In the lab, you’ll likely use a viscometer. There are a few types:
-
Capillary viscometers: Good for thin liquids.
-
Falling ball viscometers: You literally time how long a ball takes to sink.
-
Rotational viscometers: These spin a probe in the fluid to see how much resistance it meets.
Measuring the viscosity of a polymer solution can actually tell you how big the molecules are. It’s like using a stopwatch and a jar of goo to do high-level chemistry.
Final Thoughts
Mastering viscosity isn’t about being a human calculator; it’s about understanding the “why” behind the flow. For those of you aiming for IIT JAM 2027, this topic is a bridge. It connects the theory you read in books to the practical stuff engineers and chemists do every day.
If you focus on the trend—like how temperature flips the script for gases versus liquids—and get your head around the Hagen-Poiseuille law, you’ll be in a great spot for those tricky numerical answer type (NAT) questions.
To know more in detail from our expert faculty, watch our YouTube video:
Frequently Asked Questions
What are the standard units of viscosity?
In the SI system, it is measured in Pascal-seconds (Pa·s) or kg·m⁻¹·s⁻¹. In the CGS system, the unit is Poise (P). Note that 1 Pa·s = 10 Poise.
What is the difference between dynamic and kinematic viscosity?
Dynamic viscosity (μ) measures internal resistance to flow, while kinematic viscosity (ν) is the ratio of dynamic viscosity to density (ν= μ /ρ).
How does viscosity differ from density?
Density is mass per unit volume, whereas viscosity is the resistance to flow. For example, oil is less dense than water (it floats) but is more viscous (it flows more slowly).
What is a Newtonian fluid?
A Newtonian fluid is one where the viscosity remains constant regardless of the shear rate applied (e.g., water, air, alcohol).
How does temperature affect the viscosity of liquids?
As temperature increases, the kinetic energy of molecules increases, weakening intermolecular forces. Consequently, the viscosity of liquids decreases.
How does temperature affect the viscosity of gases?
In gases, viscosity is caused by molecular collisions. As temperature rises, molecular momentum transfer increases, causing the viscosity of gases to increase.
Why is honey more viscous than water?
Honey has a more complex molecular structure and stronger intermolecular forces (like hydrogen bonding), which creates higher internal friction compared to water.
Does pressure affect the viscosity of a fluid?
For most liquids, viscosity increases slightly with very high pressure. For gases, viscosity is largely independent of pressure at moderate levels.
What is the difference between laminar and turbulent flow?
Laminar flow is smooth and orderly, occurring at low Re (high viscosity). Turbulent flow is chaotic with eddies, occurring at high Re (low viscosity).
What is Poiseuille’s Law?
It describes the pressure drop in a Newtonian fluid flowing through a long cylindrical pipe. It states that the flow rate is inversely proportional to the viscosity.
What is the significance of the coefficient of viscosity?
It is the proportionality constant in Newton's Law of Viscosity, relating shear stress to the velocity gradient between fluid layers.
Why is viscosity important for IIT JAM Chemistry?
It is a core part of the "Gaseous State" and "Liquid State" chapters, used to understand molecular interactions and transport properties.
How can I avoid mistakes in NAT (Numerical Answer Type) questions?
Always ensure your units are consistent. Convert Poise to Pa·s or vice versa before plugging values into equations like the Hagen-Poiseuille formula.
What are some real-world industrial applications of viscosity?
It is critical in designing pipelines for oil transport, formulating paints, manufacturing lubricants, and optimizing HVAC systems.














