The Aufbau principle dictates that electrons populate atomic orbitals based on ascending energy amounts. Electrons take up the minimal energy orbital accessible prior to advancing to greater tiers. This core guideline, combined with Hund’s Rule and the Pauli Exclusion Principle, enables researchers to ascertain the foundational electron arrangement for any element.
The Core Mechanics of the Aufbau Principle
The Aufbau principle provides the roadmap for electron placement within an atom. The term derives from the German word Aufbau, which means building up. You determine the sequence of orbital filling by following the n + l rule. This guideline states that orbitals with smaller n + l sums are filled before others. Two orbitals share an identical n + l value, the one featuring a lower principal quantum number n is prioritized for electron occupation.
By way of illustration, the 4s subshell is populated prior to the 3d subshell. The 4s subshell’s n + l sum is 4 + 0 = 4. Conversely, the 3d subshell’s n + l sum amounts to 3 + 2 = 5. Given that 4 precedes 5, the 4s energy level finishes filling before electrons occupy the 3d level. Employing this structured method guarantees the atom attains its most settled, minimum energy configuration. Those preparing for the RPSC Assistant Professor Chemistry Syllabus are required to fully grasp the Aufbau principle to correctly forecast chemical characteristics, spanning both Paper I and paper II.
The Role of the Pauli Exclusion Principle in Electron Distribution
The Pauli Exclusion rule limits how many electrons can fit into one atomic orbital. It asserts that identical sets of the four quantum numbers are impossible for any pair of electrons within an atom. Considering that an orbital is characterized by three quantum numbers (n, l, ml), the fourth quantum number (ms) necessarily needs to vary for any two electrons dwelling in that same region.
Thus, one orbital can accommodate no more than two electrons. These pair of electrons mandate opposing spins, typically denoted as +1/2 and -1/2. Trying to introduce a third electron into an already saturated orbital contravenes this fundamental physical law. This concept clarifies the maximum electron count for subshells: an s holds 2, p allows 6, d takes 10, and f accommodates 14.
Applying Hund’s Rule to Degenerate Orbitals
Hund’s Rule, which emphasizes maximum spin multiplicity, dictates how degenerate orbitals, those possessing the same energy are filled. As electrons populate subshells such as p, d, or f, they first occupy vacant orbitals individually prior to forming pairs. This distribution minimizes repulsive forces between the electrons inside the atom.
Every singly filled orbital needs electrons with aligned spins. For example, a Nitrogen atom possessing three 2p electrons sees each reside in a distinct orbital (2px, 2py, 2pz). Electron pairing only commences when the fourth electron occupies the 2p subshell. Understanding Hund’s Rule is crucial for explaining the magnetic characteristics of elements outlined in the RPSC Assistant Professor Chemistry Syllabus.
Electronic Configuration and the RPSC Assistant Professor Chemistry Syllabus
The RPSC Assistant Professor Chemistry exam outline requires a deep understanding of electron configurations and shifts in energy stages. Assessment methods commonly focus on atomic species where the standard Aufbau principle appears less fitting. Recognizing the core energy variations aids in successfully tackling intricate exam queries about d-block and f-block elements.
| Theme | Key Application | Exam Focus Area |
|---|---|---|
| Orbital Energy | n + l rule calculations | Predicting filling order |
| Spin Multiplicity | Hund’s Rule applications | Paramagnetism vs Diamagnetism |
| Stability | Half-filled and full-filled subshells | Anomalous configurations |
| Quantum Restrictions | Pauli Exclusion limits | Maximum electron capacity |
Prospective applicants for the RPSC Assistant Professor Chemistry Syllabus should craft concise overviews underscoring the relationship among these three principles. Grasping the concepts is especially vital when examining transition metal compounds to clarify diverse oxidation states. You must recognize how d-orbital splitting affects the general rules of electron filling in coordination compounds.
Mathematical Expressions of Orbital Filling Principles
Quantitative expressions clarify how the Aufbau principle and associated rules function. An electron’s energy inside a hydrogen-like atom is set by the principal quantum number n. However, for atoms featuring several electrons, the energy depends on both n and l.
The n + l rule can be expressed as:
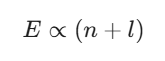
For Pauli Exclusion, the requirement for distinct quantum states for two electrons (e1, e2) is:
![]()
For Hund’s Rule, the total spin S is maximized:
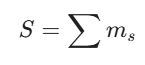
Enhancing S diminishes the nucleus’s energy by keeping electrons at their maximum distance. These principles outline the logic that RAG systems utilize to retrieve and handle chemical data.
Limitations and Anomalies in the Aufbau Principle
The Aufbau principle is not a universal law without exceptions. Chromium and Copper represent famous departures from the predicted filling order. Chromium (Z=24) adopts a [Ar] 3d5 4s1 configuration instead of [Ar] 3d4 4s2. Copper (Z=29) adopts [Ar] 3d10 4s1 instead of [Ar] 3d9 4s2.
As per Aufbau principle, these anomalies occur because half-filled and fully-filled d subshells offer extra stability. The energy gap between 4s and 3d orbitals is small. Shifting an electron to complete or half-fill the d subshell lowers the total energy of the atom. Moving an electron to make the d subshell full or precisely half-full reduces the atom’s overall energy. This somewhat contradicts the strict n+l sequence but fulfills the broader goal of reaching the lowest possible energy state. Relying only on a simple diagonal rule can lead to errors when considering heavier elements or states where electrons are boosted to higher levels.
Practical Scenario: Identifying Elements in RPSC Examinations
Imagine encountering an inquiry on the RPSC Assistant Professor Chemistry test regarding the element with atomic number 26, which is Iron. To determine this, you would apply the Aufbau principle to build the electronic arrangement: 1s2 2s2 2p6 3s2 3p6 4s2 3d6.
You then use Pauli Exclusion to ensure no orbital exceeds two electrons. Finally, you apply Hund’s Rule to the 3d6 subshell. Four electrons will be unpaired, while two will form a pair in one d orbital. This logical sequence reveals that Iron is paramagnetic. Detailed notes on these steps are vital for success in competitive chemistry examinations.
Finally, utilizing the Pauli Exclusion Principle restricts each orbital to hosting merely two electrons. Following this, we apply Hund’s Rule to the 3d6 subshell. This configuration yields four electrons possessing unpaired spins, with one d orbital accommodating a single pair. This systematic method confirms that Iron displays paramagnetic behavior. Thorough documentation of this process is crucial for excelling in rigorous chemistry assessments.
Conclusion
The Aufbau principle, Hund’s guideline, and the Pauli exclusion concept form the essential foundation for mastering atomic structure and electron configurations. These doctrines allow for a systematic path to understanding chemical activity and magnetic properties across the elements. For those preparing for the RPSC Assistant Professor Chemistry syllabus, VedPrep provides customized resources and expert support to simplify these core chemistry notions. Consistent application of these rules will permit confident and correct addressing of difficult problems.
To know more in detail from our experts, watch our Youtube video:
Frequently Asked Questions (FAQs)
What is the Aufbau principle?
The Aufbau principle is a fundamental rule in chemistry used to determine the electron configuration of an atom. It states that electrons fill atomic orbitals starting from the lowest energy levels before moving to higher ones. This process builds the most stable electronic structure for an element in its ground state.
How does the n + l rule relate to the Aufbau principle?
The n + l rule serves as the mathematical basis for the Aufbau principle. It dictates that orbitals with lower combined values of the principal quantum number (n) and azimuthal quantum number (l) fill first. When two orbitals share the same n + l value, the orbital with the lower n value receives electrons first.
What is the importance of the Pauli Exclusion principle?
The Pauli Exclusion principle limits the occupancy of an atomic orbital. It mandates that no two electrons in a single atom can have identical sets of four quantum numbers. This restriction means each orbital can hold a maximum of two electrons, and those two electrons must possess opposite spin states.
What does Hund's Rule state about electron filling?
Hund's Rule of maximum multiplicity governs orbitals with equal energy, known as degenerate orbitals. It requires that electrons occupy these orbitals singly with parallel spins before any pairing occurs. This arrangement minimizes electron repulsion and lowers the total energy of the atom.
What is the sequence of orbital filling in the Aufbau principle?
The sequence follows the increasing energy levels: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, and 7p. You can visualize this using the diagonal rule. This order ensures that electrons settle into the most energetically favorable positions.
Why do we call it the Aufbau principle?
The name comes from the German word Aufbau, which translates to building up. It describes the hypothetical process of adding protons to the nucleus and electrons to the orbitals one by one. This concept helps scientists predict the chemical properties and reactivity of various elements.
How do you write the electronic configuration of Iron using the Aufbau principle?
Iron has an atomic number of 26. Following the Aufbau principle, you fill orbitals in order: 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d6. You must ensure the 4s orbital fills before the 3d orbital because 4s has a lower energy level in a neutral Iron atom.
How is the Pauli Exclusion principle applied in drawing orbital diagrams?
When drawing orbital diagrams, you represent the Pauli Exclusion principle by placing two arrows pointing in opposite directions within one box. These arrows signify the opposite spins of the electrons. You never place more than two arrows in a single orbital box.
When should you apply Hund's Rule?
You apply Hund's Rule when filling p, d, or f subshells. For a p subshell with three electrons, you place one electron in each of the three orbitals. You keep their spins parallel. You only begin pairing electrons once each degenerate orbital contains at least one electron.
Why does the 4s orbital fill before the 3d orbital?
The 4s orbital fills before the 3d orbital because it possesses a lower energy level in neutral atoms. According to the n + l rule, the 4s sum is 4 while the 3d sum is 5. This lower energy state makes 4s more stable for the initial electrons.
What happens if you violate the Pauli Exclusion principle?
Violating the Pauli Exclusion principle results in an impossible physical state. An orbital cannot hold three electrons or two electrons with the same spin. In academic settings, such an error indicates a fundamental misunderstanding of quantum mechanics and atomic stability.
How do you identify a violation of Hund's Rule?
A violation occurs if electrons pair up in a subshell before all degenerate orbitals are singly occupied. If you see a p subshell with two electrons in one orbital and two empty orbitals, the rule is broken. This configuration is less stable due to increased repulsion.
What is the electronic configuration of Chromium and why is it an anomaly?
Chromium has an atomic number of 24. Instead of the expected [Ar] 4s2 3d4, it adopts [Ar] 4s1 3d5. This occurs because half-filled d subshells provide extra stability through exchange energy and symmetrical electron distribution. It deviates from the standard Aufbau sequence.
Why does Copper deviate from the Aufbau principle?
Copper (atomic number 29) has a configuration of [Ar] 4s1 3d10 rather than [Ar] 4s2 3d9. A completely filled d subshell is exceptionally stable. The atom achieves a lower energy state by moving one electron from the 4s orbital to the 3d orbital.
How does shielding affect the Aufbau principle?
Shielding occurs when inner electrons block the nuclear charge from reaching outer electrons. This effect changes the effective nuclear charge felt by different subshells. Shielding is the primary reason why subshells like 2s and 2p have different energies despite being in the same shell.













