Fission and Fusion represent the two main nuclear reactions that let loose huge energy stores by changing the structure of atomic cores. Fission involves dividing a large nucleus into smaller pieces, whereas fusion joins light nuclei to form a larger one. These two processes are fundamental to the fields of Nuclear and Particle Physics and are crucial subjects for the RPSC Assistant Professor Physics Paper II exam.
Core Mechanisms of Nuclear Fission
Regarding Fission and Fusion, nuclear fission takes place when a large nucleus, like Uranium 235, turns unstable and divides into two or more smaller nuclei. This action generally starts once the nucleus captures a neutron. The resulting pieces have a total mass smaller than the initial nucleus. This lost mass transforms into a tremendous amount of energy. You can see this effect in nuclear power plants where managed chain reactions produce electrical power.
Within the domain of Nuclear and Particle Physics, the process of fission hinges upon the binding energy metric per nucleon. Heavier atomic nuclei exhibit diminished stability as their binding energy per nucleon falls beneath that characteristic of elements positioned centrally in the periodic table. Upon nuclear cleavage, the resulting fragments progress towards a more stable configuration. This shift results in an energetic discharge, consistent with Einstein’s principle of mass-energy equivalence. Candidates preparing for RPSC Assistant Professor Physics Paper II must master the liquid drop model to explain these nuclear deformations.
Principles of Nuclear Fusion
Nuclear fusion is the process where two light atomic nuclei combine to form a single heavier nucleus as per Fission and Fusion. This process fuels stellar bodies, the Sun among them. For fusion to happen, atomic nuclei need to surmount the intense electrostatic repulsion present between protons, which carry a positive charge. This necessitates exceptionally high heat and pressure to get the nuclei sufficiently near for the powerful nuclear force to engage. Fusion yields substantially more energy for a given mass compared to fission.
Research in Nuclear and Particle Physics focuses on achieving controlled fusion on Earth. Hydrogen isotopes like Deuterium and Tritium are the most common fuels for these reactions. The energy emission arises because the mass of the resultant helium atom is less than the total weight of the precursor hydrogen atoms. Understanding the Coulomb barrier and quantum tunneling phenomena is crucial for tackling questions in the RPSC Assistant Professor Physics Paper II.
Mathematical Foundations of Fission and Fusion
The energy released in these reactions is quantifiable through specific mathematical expressions. The Q value represents the net energy released during a nuclear transformation. In both Fission and Fusion, you calculate the energy by finding the difference between the rest masses of the reactants and the products. This calculation is a frequent requirement for students tackling the RPSC Assistant Professor Physics Paper II.
The following table provides the essential formulas used to calculate the energetics in Nuclear and Particle Physics.
| Process | Mathematical Expression | Variables Defined |
|---|---|---|
| Mass Defect | Z=Atomic Number, A=Mass Number, m=Mass | |
| Energy Release (Q-Value) | M=Rest Mass, c=Speed of Light | |
| Fission Energy Example | 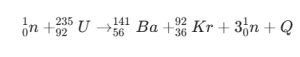 |
Standard Uranium 235 fission reaction |
| Fusion Energy Example | Deuterium-Tritium fusion reaction |
Comparison of Energy Yield and Efficiency
Fission and Fusion diverge significantly in terms of energy concentration and ease of obtaining fuel. Fission relies on weighty materials such as Uranium or Plutonium, which are limited supplies. This process generates waste that remains radioactive for extended periods, necessitating enduring storage arrangements. Despite this, fission apparatus is well-developed and presently furnishes a consistent stream of emissions-free baseline electricity worldwide. You will find that most current syllabus requirements for RPSC Assistant Professor Physics Paper II emphasize the efficiency calculations of these power cycles.
Based on Fission and Fusion, Fusion offers the potential for nearly limitless energy using isotopes found in seawater. It produces no long lived radioactive waste and carries no risk of a meltdown. The challenge lies in the confinement of plasma at millions of degrees Celsius. Within the study of Nuclear and Particle Physics, the Lawson criterion defines the conditions needed for a fusion reactor to reach ignition. Understanding these constraints is essential for advanced academic examinations.
Critical Perspective on Stability and the Iron Peak
A common misconception is that all nuclei can undergo either Fission or Fusion to release energy. This is incorrect. The curve of binding energy shows that stability peaks at Iron 56. Substances less massive than iron emit power via fusion, whereas elements exceeding iron in mass release energy through splitting. Iron itself represents the most steadfast nucleus and is unable to yield power by either method under typical circumstances.
This constraint clarifies why stellar nucleosynthesis ceases at Iron within average stars. After a star’s center becomes Iron, fusion no longer supplies the outward force needed to balance gravity, resulting in a supernova. If you are preparing for RPSC Assistant Professor Physics Paper II, you must distinguish between exothermic and endothermic nuclear reactions. Forcing fusion in elements heavier than Iron requires an input of energy rather than releasing it.
Applications in Modern Science and Industry
Fission and Fusion have uses extending well past energy production. In Nuclear and Particle Physics, fragments from fission act as origins for medical isotopes employed in diagnosing and treating cancer. Neutron scattering methods, stemming from fission emitters, enable researchers to examine material composition at the atomic scale. Such real-world examples form essential parts of the RPSC Assistant Professor Physics Paper II syllabus.
Research in Fission and Fusion spurs progress in areas like high-temperature substances and superconducting magnets. A tangible use involves creating fusion drive systems for extended voyages through space. VedPrep consistently helps students achieve AIR 1s and top rankings annually across Physics, Chemistry, Biology, and Mathematics.
Conclusion
Exploring Nuclear and Particle Physics continues to be fundamental for grasping the cosmos’s energetic movements. Splitting Fission and Fusion are more than just classroom ideas; they are the processes powering both the development of stars and worldwide energy sources. Though fission offers quick, adaptable power by moving heavy atomic cores, fusion presents the future prospect of enduring, high-output electricity via the merging of light atomic cores. Achieving success on the RPSC Assistant Professor Physics Paper II demands an accurate comprehension of these power shifts and their corresponding mathematical structures.
As investigations advance in Fission and Fusion, controlling these reactions will shape the coming era of industrial and healthcare applications. Whether scrutinizing the stability around the iron peak or determining Q-values, learners need to engage these subjects with sharp analytical skills. VedPrep provides focused preparation and top-tier materials, guaranteeing applicants secure high placements in the RPSC Assistant Professor test.
To learn more in detail from our expert professional, watch our YouTube video:
Frequently Asked Questions (FAQs)
What is the difference between fission and fusion?
Fission occurs when a heavy atomic nucleus splits into two or more smaller nuclei. Fusion happens when two light nuclei combine to form a heavier one. Both processes release energy due to mass defect. Fission is currently the standard for power plants. Fusion is the energy source for stars.
How does nuclear fission release energy?
Fission releases energy when a heavy nucleus becomes unstable after absorbing a neutron. The nucleus splits into smaller fragments with less total mass than the original atom. This lost mass converts into energy through the principle of mass energy equivalence. This process produces high velocity neutrons and heat.
Why does fusion require high temperatures?
Nuclear fusion requires high temperatures to overcome the electrostatic repulsion between positively charged nuclei. Atoms must move at extreme speeds to get close enough for the strong nuclear force to bind them. On Earth, scientists use plasma heated to millions of degrees to achieve these conditions.
What are the common fuels used in fission and fusion?
Fission reactors primarily use heavy elements like Uranium 235 or Plutonium 239. These isotopes are fissile and maintain a chain reaction. Fusion research focuses on hydrogen isotopes like Deuterium and Tritium. Deuterium is abundant in seawater. Tritium is typically bred from lithium during the nuclear reaction.
How is fission controlled in a nuclear reactor?
Engineers control fission using control rods made of neutron absorbing materials like Boron or Cadmium. Sliding these rods into the reactor core slows the chain reaction by removing available neutrons. Moderators like water or graphite also slow down fast neutrons to ensure they cause further fission events efficiently.
What methods achieve plasma confinement for fusion?
Scientists use magnetic confinement and inertial confinement to contain fusion plasma. Magnetic confinement uses powerful superconducting magnets in a donut shaped device called a Tokamak. Inertial confinement uses high power lasers to compress fuel pellets. Both methods aim to meet the Lawson criterion for a self sustaining reaction.
Why do some chain reactions become uncontrolled?
A chain reaction becomes uncontrolled if the number of neutrons causing new fissions exceeds a critical threshold. This happens when the multiplication factor is greater than one without proper regulation. Modern reactor designs use passive safety systems to prevent such surges. Overheating can lead to fuel damage or containment failure.
What are the main challenges in commercializing fusion?
Commercial fusion faces hurdles in sustaining high temperature plasma for long durations. Materials inside the reactor must withstand intense neutron bombardment without degrading. Capturing the released energy and converting it into electricity efficiently is also a major engineering task. Economic viability remains a significant hurdle for current experimental designs.
How is radioactive waste managed in fission?
Fission waste requires specialized storage due to its long half life. Short lived isotopes are stored in cooling pools at the reactor site. Long lived high level waste is vitrified into glass and placed in deep geological repositories. This prevents radioactive materials from entering the biosphere over thousands of years.
Why is Uranium 238 not used for thermal fission?
Uranium 238 is a fertile isotope rather than a fissile one. It requires high energy neutrons to undergo fission. Thermal neutrons are usually absorbed by Uranium 238 to form Plutonium 239 instead. Most reactors use enriched uranium to increase the concentration of Uranium 235 for better performance.
What is the significance of the Iron Peak in nucleosynthesis?
The Iron Peak represents the limit of energy production through stellar fusion. Iron has the most stable nucleus, meaning fusing iron requires more energy than it releases. Once a massive star forms an iron core, energy production ceases. This leads to the gravitational collapse and subsequent supernova explosion of the star.
How does tunneling contribute to nuclear fusion in stars?
Quantum tunneling allows nuclei to overcome the Coulomb barrier even when they lack sufficient kinetic energy for classical fusion. High pressure in stellar cores increases the probability of these tunneling events. This process allows stars to burn hydrogen at lower temperatures than would be required by classical physics alone.
What are the advantages of thorium based fission?
Thorium is more abundant than uranium and produces less long lived transuranic waste. Thorium 232 is fertile and converts into fissile Uranium 233 upon neutron absorption. Thorium reactors can operate in a molten salt configuration for enhanced safety. This technology offers a potential alternative to traditional pressurized water reactors.
How does neutron radiation affect reactor materials?
Neutron radiation causes atoms in structural materials to displace from their lattice positions. This leads to swelling, embrittlement, and loss of mechanical strength over time. Materials science research focuses on developing alloys that resist this radiation damage. Understanding these effects is vital for extending the lifespan of nuclear power plants.













