Isomerism refers to the existence of two or more compounds with the same molecular formula but different arrangements of atoms. These compounds, known as isomers, possess distinct physical or chemical properties because of their unique structural or spatial orientations. Understanding Isomerism is fundamental for mastering organic synthesis, drug design, and chemical reactivity.
Structural Isomerism and Its Molecular Classifications
Structural Isomerism occurs when atoms within a molecule connect in different sequences despite sharing the same molecular formula. This category relies on the connectivity of the carbon skeleton or the position of functional groups. You will find that these differences significantly alter boiling points, melting points, and chemical stability.
Chain isomers differ in the arrangement of the carbon backbone. For example, pentane exists as a straight chain; whereas, isopentane features a branched structure. Positional isomers maintain the same carbon skeleton but vary the location of a functional group or substituent. In propanol, the hydroxyl group sits on the first or second carbon, creating two distinct molecules. Functional isomers possess the same formula but belong to different homologous series, such as ethanol and dimethyl ether. Metamerism arises from unequal distribution of alkyl groups around a polyvalent functional group like an ether or ketone. Tautomerism involves a dynamic equilibrium between two forms, typically the keto and enol structures, through the migration of a hydrogen atom and a double bond.
Stereoisomerism and Spatial Orientation
Stereoisomerism involves molecules with identical connectivity but different three dimensional orientations. This branch of chemistry divides into geometrical and optical categories. You must analyze the spatial arrangement to predict how these molecules interact with plane polarized light or biological receptors.
Optical isomers or enantiomers are non superimposable mirror images. They contain at least one chiral center, which is a carbon atom bonded to four different groups. Diastereomers are stereoisomers that are not mirror images of each other. These differences dictate how a drug fits into a protein binding site. Many pharmaceutical compounds rely on specific stereoisomers for efficacy; whereas, the wrong isomer might cause toxicity.
Geometrical Isomerism in Alkenes and Cyclic Compounds
Geometrical Isomerism results from restricted rotation around a double bond or within a ring structure. This restriction creates two distinct forms: cis and trans. The E/Z system provides a more precise nomenclature when four different groups attach to the double bond. Candidates must focus on Isomerism syllabus to cover key areas of RPSC Assistant Professor Chemistry.
In the E/Z system, you assign priority to groups based on the Cahn Ingold Prelog rules. High priority groups on the same side of the double bond constitute the Z isomer. High priority groups on opposite sides form the E isomer. For cyclic compounds, substituents can sit on the same side or opposite sides of the ring plane. This spatial fixedness dictates the physical properties of the molecule.
| Isomer Type | Configuration Description | Example Compound |
|---|---|---|
| Cis | Similar groups on the same side | Cis-2-butene |
| Trans | Similar groups on opposite sides | Trans-2-butene |
| Z (Zusammen) | High priority groups on same side | (Z)-1-bromo-2-chloroethene |
| E (Entgegen) | High priority groups on opposite sides | (E)-1-bromo-2-chloroethene |
Stereoisomerism and the R/S System of Nomenclature
The R/S system identifies the absolute configuration of a chiral center. You must rank the four substituents by atomic number. Looking down the bond from the chiral center to the lowest priority group, you determine the sequence of the remaining three groups.
Clockwise sequences indicate the R configuration. Counterclockwise sequences indicate the S configuration. This systematic approach eliminates ambiguity found in older naming conventions. You apply these rules to distinguish between molecules like (R)-lactic acid and (S)-lactic acid. Understanding this system is vital for interpreting Previous Year Questions (PYQ) Isomerism trends in competitive examinations.
Conformational Analysis of Alkenes and Cycloalkanes
Conformational Analysis examines the different spatial arrangements produced by rotation around single sigma bonds. Unlike other types of Isomerism, conformers interconvert rapidly at room temperature. You measure the energy differences between these states to determine the most stable form.
In ethane, the staggered conformation represents the lowest energy state because it minimizes electron repulsion. The eclipsed conformation is the highest energy state due to torsional strain. For butane, the anti conformation is more stable than the gauche form because of reduced steric hindrance between methyl groups.
Cycloalkanes exhibit unique strains including angle strain and Newman projection eclipsing. Cyclohexane avoids angle strain by adopting a chair conformation. The chair form places all C-H bonds in staggered positions, making it the most stable arrangement. Axial and equatorial positions in the chair form determine the stability of substituted cyclohexanes. Large groups prefer equatorial positions to avoid 1,3-diaxial interactions.
Asymmetric Synthesis and Reaction Selectivity
Asymmetric Synthesis focuses on the production of a specific enantiomer or diastereomer. In traditional reactions, achiral starting materials often yield a racemic mixture. Stereoselective reactions favor the formation of one stereoisomer over others. Stereospecific reactions dictate that the stereochemistry of the reactant determines the stereochemistry of the product.
You use chiral catalysts or auxiliaries to direct the outcome of these reactions. For example, the hydrogenation of a pro-chiral alkene using a chiral catalyst produces a single enantiomer. These techniques are essential in the synthesis of complex natural products and modern medicines.
Previous Year Questions (PYQ) Isomerism and Numerical Applications
Analyzing Previous Year Questions (PYQ) Isomerism reveals a focus on calculating the number of possible isomers. You must apply specific formulas to determine the quantity of structural and optical isomers for a given formula.
The number of optical isomers for a molecule with ‘n’ chiral centers and no symmetry is 2n. If the molecule possesses internal symmetry, you use modified equations to account for meso compounds. Meso compounds contain chiral centers but are achiral overall due to an internal plane of symmetry.
| Case | Formula for Total Stereoisomers |
|---|---|
| No symmetry, n chiral centers | 2n |
| Symmetrical, even n | 2n-1 + 2(n/2)-1 |
| Symmetrical, odd n | 2n-1 |
Numerical problems often require you to calculate the specific rotation of a mixture. You use the formula:
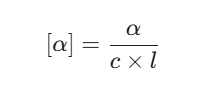
Where [α] is specific rotation, α is observed rotation, c is concentration in g/mL, and l is path length in decimeters.
Limitations of Standard Isomerism Models
A common misconception suggests that all molecules with chiral centers are optically active. This belief fails when considering meso compounds. Despite having multiple chiral centers, these molecules do not rotate plane polarized light because their internal symmetry cancels out the individual rotations.
Another limitation occurs in Conformational Analysis. While textbooks often treat conformers as transient, certain bulky groups can create atropisomers. These are conformers that cannot interconvert due to high rotational barriers, making them isolable stereoisomers at room temperature. You must identify when steric bulk overrides the assumption of free rotation.
Practical Application in Pharmaceutical Development
The significance of Isomerism is most evident in the thalidomide tragedy. One enantiomer provided sedative effects; whereas, the other caused birth defects. Modern drug development now requires the separation and testing of individual enantiomers.
In your work with chemical synthesis, you prioritize stereospecific pathways to ensure product purity. The production of L-Dopa for Parkinson’s disease relies on asymmetric catalysis to ensure only the active isomer reaches the patient. This precision reduces dosages and minimizes side effects.
Conclusion
Mastering the complexities of Isomerism is a vital step for any chemist or student aiming to excel in molecular sciences. By distinguishing between structural connectivity and spatial orientation, you gain the ability to predict molecular behavior and reactivity in diverse chemical environments. VedPrep provides comprehensive resources and expert guidance to help you navigate these advanced concepts with ease. Continuous practice with these naming systems and conformational models ensures you can tackle challenging problems in both academic and professional settings.
To learn from our top faculty, watch this Youtube Video,
Frequently Asked Questions (FAQs)
What is Isomerism in organic chemistry?
Isomerism describes molecules with the same molecular formula but different atomic arrangements or spatial orientations. These variations result in distinct physical and chemical properties. You categorize isomers into structural isomers, which differ in connectivity, and stereoisomers, which differ in three dimensional arrangement.
How does Structural Isomerism differ from Stereoisomerism?
Structural Isomerism involves atoms bonded in different sequences. You will see different carbon skeletons or functional group positions. Stereoisomerism maintains identical bonding sequences but varies how atoms occupy space. This distinction determines whether a molecule interacts with specific biological receptors or rotates plane polarized light.
What are the primary types of Structural Isomerism?
Structural Isomerism includes chain, positional, functional, metamerism, and tautomerism. Chain isomers vary in the carbon backbone. Positional isomers change the location of a functional group. Functional isomers have different functional groups altogether. Metamerism involves different alkyl distribution, while tautomerism is a dynamic equilibrium between two forms.
Why is Geometrical Isomerism classified as a stereoisomer?
Geometrical Isomerism qualifies as stereoisomerism because the atoms connect in the same order but differ in spatial orientation. This occurs due to restricted rotation around double bonds or rings. You identify these as cis and trans or E and Z forms based on the priority of attached groups.
How do you assign E and Z configurations to an alkene?
You rank the two groups on each carbon of the double bond using atomic numbers. If the two high priority groups sit on the same side, the configuration is Z. If they sit on opposite sides, the configuration is E. This system provides more precision than the cis trans method.
How do you determine the number of stereoisomers for a molecule?
You identify the number of chiral centers, denoted as n. For a molecule without internal symmetry, the total number of stereoisomers is 2 to the power of n. If the molecule is symmetrical, you must subtract meso compounds from this total to find the correct value.
How do you identify meso compounds in Isomerism problems?
A meso compound contains multiple chiral centers but remains achiral. You look for an internal plane of symmetry that divides the molecule into two identical halves. This symmetry causes the optical rotation of one half to cancel the other, resulting in zero net rotation.
How do you solve Previous Year Questions (PYQ) Isomerism problems efficiently?
You should first determine the degree of unsaturation to narrow down possible structural formulas. Then, identify any chiral centers or restricted bonds to account for stereoisomers. Categorizing the question into structural or spatial types prevents common errors in counting total isomers.
Why does a racemic mixture show no optical activity?
A racemic mixture contains equal amounts of two enantiomers. One enantiomer rotates light in a clockwise direction, and the other rotates it counterclockwise by the same degree. These opposite effects cancel each other out, making the solution appear optically inactive to a polarimeter.
How do you distinguish between enantiomers and diastereomers?
Enantiomers are exact mirror images of each other at every chiral center. Diastereomers are stereoisomers that are not mirror images. This happens when at least one, but not all, chiral centers in a molecule change their configuration.
What causes the instability of the eclipsed conformation in alkanes?
The eclipsed conformation suffers from torsional strain and steric hindrance. Atoms or lone pairs on adjacent carbons are forced into close proximity, increasing electron repulsion. You find that the staggered conformation is more stable because it maximizes the distance between these groups.
What is the role of Asymmetric Synthesis in drug manufacturing?
Asymmetric Synthesis uses chiral catalysts to produce only one specific enantiomer. This process is crucial because different enantiomers can have vastly different biological effects. You use this to ensure medicine is both safe and effective while reducing chemical waste.
What are atropisomers in the context of Stereoisomerism?
Atropisomers are conformers that become stable isomers because rotation around a single bond is physically blocked. This usually occurs in bulky substituted biphenyls. If the energy barrier to rotation is high enough, you can isolate these individual forms at room temperature.
How does Tautomerism differ from Resonance?
Tautomerism involves the actual movement of an atom, usually hydrogen, and the relocation of a pi bond. It creates two distinct molecules in equilibrium. Resonance involves only the delocalization of electrons within a single molecular structure and does not change atom positions.
How do you apply Conformational Analysis to substituted cycloalkanes?
You must consider the size of the substituent. Large groups like tert butyl prefer the equatorial position to avoid 1,3-diaxial interactions with hydrogens on the ring. Placing bulky groups in the axial position creates significant steric strain and raises the molecular energy.














