The Kinetic Molecular Theory of gases is the theoretical framework that bridges the gap between individual molecular behavior and the macroscopic properties of gases like pressure, volume, and temperature. For IIT JAM 2027 aspirants, this concept is non-negotiable as it serves as the foundation for both Physical Chemistry and Thermal Physics. By treating gas molecules as point masses in constant, chaotic motion, the KMT provides a mathematical derivation for the Ideal Gas Law (PV = nRT) and explains how energy is distributed within a system.
Syllabus – Thermodynamics and Statistical Mechanics (Unit 2)
Despite its foundational role in IIT JAM 2027, the treatment of gas behavior differs slightly between assessments in IIT NET syllabus. Where one emphasizes velocity distributions, the other builds toward broader probabilistic frameworks. With attention to molecular motion, early study sections introduce assumptions about particle collisions. Following that, energy partitioning appears across problem sets in varied forms. Although derived from similar principles, each exam structures questions around distinct applications. Through layered complexity, statistical ideas emerge beyond basic kinetic equations.
Standard academic references for this unit include:
“Physical Chemistry” by P.W. Atkins: Excellent for understanding the derivation of collision frequency and mean free path.
“Thermodynamics” by C. J. Adkins: A classic for grasping the heat and work relationship through molecular motion.
“Statistical Mechanics” by R. K. Pathria: Essential for those transitioning from JAM to GATE/NET levels, focusing on the partition functions.
For IIT JAM 2027, the syllabus specifically emphasizes the derivation of the pressure equation, the concept of degrees of freedom, and the Law of Equipartition of Energy.
Core Concept: Kinetic Molecular Theory of Gases For IIT JAM
At its heart, the Kinetic Molecular Theory is a model that simplifies the complex interactions of real gases into a set of predictable “ideal” behaviors.
Molecular Motion and Pressure
The theory posits that gas molecules are in constant random motion. Gas molecules move with sufficient thermal energy, unlike those in solids or liquids, separating entirely due to weakened intermolecular forces. From the perspective of Kinetic Molecular Theory, what emerges is pressure – defined not as fixed push but as combined effect of countless molecule-wall impacts occurring elastically. With every strike against the surface, momentum shifts slightly; such repeated alterations, tracked across space and time, form the observable value called pressure. Though invisible individually, these interactions collectively yield measurable outcomes under consistent conditions.
Temperature and Kinetic Energy
It becomes clear, upon studying thermodynamics, how closely linked temperature is to molecular motion. What appears as heat externally reflects internal particle movement on a tiny scale. A rise in reading on any thermometer signals faster jiggling of atoms inside matter. This connection shows why cooling slows down particles until they barely vibrate. Temperature, then, stands as nothing more than an overall indicator of such activity across countless molecules.
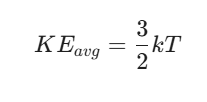
Where:
k is the Boltzmann constant (1.38 × 10-23 J/K)
T is the absolute temperature in Kelvin.
Kinetic molecular theory of gases For IIT JAM
Understanding the Kinetic Molecular Theory of Gases requires full familiarity with its four core assumptions. Such ideas frequently appear in concept-based MCQs and MSQs on the IIT JAM 2027 test. Though abstract, these principles form a foundation that shapes how gas behavior is analyzed. Questions built around them tend to focus less on calculation, more on interpretation. Each assumption acts like a lens, clarifying specific macroscopic properties through microscopic motion. Because reasoning matters here, rote memorization brings limited benefit.
Postulate 1: Composition of Gases
Most gases consist of countless minuscule units known as molecules – atoms, when referring to noble gases. Far apart relative to their dimensions, these units occupy space where gaps dominate over substance. Hence, one considers the true volume of such particles insignificant.
Postulate 2: Molecular Motion
Molecules are in ceaseless, random, straight-line motion. They do not follow curved paths unless acted upon by a collision. This randomness ensures that the gas occupies the entire volume of its container uniformly.
Postulate 3: Molecular Volume
The volume of the individual gas molecules is considered zero (point masses) relative to the total volume of the gas. Based on Kinetic Molecular Theory of Gases, this is why gases are highly compressible. For IIT JAM 2027, remember that this postulate is what fails in Real Gases, leading to the “b” constant in the van der Waals equation.
Postulate 4: Elastic Collisions
Collisions between molecules and with the container walls are perfectly elastic. This means there is no net loss of kinetic energy during a collision; energy is simply transferred from one molecule to another.
Worked Example: CSIR NET Style Question
Question: A sample of Helium gas is kept in a 10L container at 300 K. If the temperature is quadrupled to 1200K, what happens to the Root Mean Square (RMS) speed of the molecules?
Solution for IIT JAM 2027 Aspirants:
The RMS speed (vrms) is derived from KMT as:
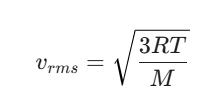
Since the molar mass (M) and Gas Constant (R) remain constant:
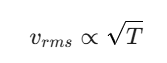
If the temperature T becomes 4T, then the new speed v’ becomes:
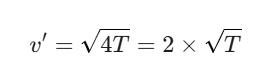
Misconception: Common Errors in Understanding Kinetic Molecular Theory of Gases
Macroscopic vs. Microscopic Motion: Students often think that if a gas is “still” in a balloon, the molecules are still. In reality, the molecules are moving at hundreds of meters per second; only the bulk velocity is zero.
Energy vs. Speed: A common error is assuming all molecules in a gas move at the same speed. They do not. They follow the Maxwell-Boltzmann distribution, where some are slow, some are very fast, and most are in the middle.
The “Negligible Volume” Trap: Students forget that this is only an assumption for ideal gases. In IIT JAM 2027 problems involving high pressure, you must account for the actual volume of the molecules (Real Gas behavior).
Application: Real-World Applications of Kinetic Molecular Theory of Gases For IIT JAM
The KMT isn’t just a textbook theory; it dictates how our modern world functions in Kinetic Molecular Theory of Gases.
Scuba Diving and Dalton’s Law: As a diver goes deeper, the pressure increases. KMT explains that the number of molecular collisions per second increases, forcing more nitrogen into the blood. Understanding partial pressures (a derivative of KMT) is life-saving for divers.
Aerospace Engineering: In designing high-altitude balloons or spacecraft, engineers use KMT to predict how the thinning atmosphere (fewer molecular collisions) will affect the internal pressure of the vessel.
Chemical Synthesis: In the Haber-Bosch process for ammonia, knowing how temperature increases molecular collision frequency allows chemical engineers at firms and research labs to optimize the rate of reaction.
Kinetic molecular theory of gases For IIT JAM
As you prepare for IIT JAM 2027, your strategy should be two-fold: conceptual clarity and numerical speed. The KMT is the bridge to Van der Waals forces, Critical Phenomena, and Liquefaction of Gases.
Preparation Tips for IIT JAM 2027:
Derive the Equation: Don’t just memorize PV = 1/3nmv2. Learn how it is derived from the change in momentum.
Master the Speeds: Be comfortable switching between vrms, vaverage, and vmost probable.
vrms : vavg : vmp = √3 : √8/π : √2
Graph Analysis: IIT JAM 2027 will likely feature graphs showing how the Maxwell distribution shifts with temperature or molar mass.
VedPrep delivers targeted instruction across defined areas. Rigor matches what appears necessary for JAM and CUET PG success. One emphasis lands on the Law of Equipartition of Energy – specifically f/2 times kT – a segment likely prominent in 2027 testing.
Final Thoughts
Though unseen, the movement of gas particles underlies consistent large-scale patterns described by the Kinetic Molecular Theory of Gases, forming a key foundation for IIT JAM 2027 preparation. Despite simple starting principles, difficulty arises when using them to explain actual gas performance, especially once conditions shift beyond ideal settings. For tests such as IIT JAM, CSIR NET, and GATE, progress comes less from repeating equations and more from grasping how speed, force, and heat relate inside collections of molecules.
Beginning with clear guidance, VedPrep offers organized support alongside focused materials to address key misunderstandings, transforming abstract ideas into accurate test performance. While progressing through study tasks, consider this: mastery of the Kinetic Molecular Theory of Gases extends beyond examination success – such understanding forms the core outlook needed for future work in chemistry-related fields.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions (FAQs)
What is the primary objective of the Kinetic Molecular Theory?
The KMT provides a microscopic model to explain the macroscopic properties of gases, such as pressure, volume, and temperature, based on the motion of individual molecules.
What are the 4 main postulates of the Kinetic Molecular Theory?
The four main postulates are: (1) Gases consist of tiny particles in constant random motion; (2) The volume of particles is negligible; (3) Intermolecular forces are non-existent; (4) Collisions are perfectly elastic.
Why is KMT important for IIT JAM 2027 aspirants?
It is a foundational topic in Physical Chemistry and Thermal Physics. Mastery of KMT is essential for solving problems in Thermodynamics, Statistical Mechanics, and Real Gas behavior.
What is meant by an "Elastic Collision" in KMT?
An elastic collision is one where there is no net loss of total kinetic energy. Energy can be transferred between molecules, but the total energy of the system remains constant.
How does KMT define gas pressure?
Pressure is defined as the force exerted by gas molecules per unit area of the container walls due to constant, high-speed collisions.
What is the relationship between temperature and kinetic energy?
According to KMT, the average kinetic energy of gas molecules is directly proportional to the absolute temperature (KEavg = 3/2kT).
Does the identity of the gas affect the average kinetic energy?
No. At a given temperature, all ideal gas molecules have the same average kinetic energy, regardless of their mass or identity.
Why do we assume gas molecules have "negligible volume"?
This assumption simplifies the math of the Ideal Gas Law (PV=nRT) by treating molecules as point masses, which is accurate at low pressures and high temperatures.
When does the Kinetic Molecular Theory fail?
KMT fails under high pressure and low temperature, where the actual volume of molecules and intermolecular attractive forces (Van der Waals forces) become significant.
How does molecular mass affect molecular speed?
At a constant temperature, heavier molecules move slower on average than lighter molecules, following the relationship v ∝ 1/√M.
What is the "Mean Free Path" in KMT?
The mean free path is the average distance a molecule travels between two successive collisions.
How does increasing temperature affect the Maxwell-Boltzmann distribution?
Increasing the temperature flattens the curve and shifts the peak (most probable speed) toward higher velocities.
What is the significance of the Boltzmann Constant (k) in KMT?
The Boltzmann constant (k = R/NA) relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas.
What are "Degrees of Freedom"?
Degrees of freedom refer to the number of independent ways a molecule can possess energy (translational, rotational, and vibrational).
How many degrees of freedom does a monatomic gas have?
A monatomic gas (like Helium) has 3 translational degrees of freedom.













