Among topics relevant to IIT JAM, Atomic spectra stand apart – not just another section in course material, but foundational to both modern physics and physical chemistry. With focus shifting toward the 2027 exam cycle, insight into how atoms emit or absorb electromagnetic waves during electron shifts becomes vital. Success at institutions like IITs or NITs often depends on mastery here. Though subtle, these patterns reveal much about energy levels when examined closely.
Light patterns from atoms appear when electrons shift among fixed energy steps. Within an atom, particles remain in set positions where energy values are distinct and limited. Excitation caused by heat or electric flow pushes an electron upward; later it falls back down. Each descent emits a light particle – the amount of energy matching exactly what was gained earlier. Because spacing between stages differs per element, colored marks on screens differ too. These markings serve like identity tags found in starlight or materials tested nearby. No two elements repeat the exact arrangement seen through such traces left behind. Matching these reveals which building blocks exist within distant suns or containers here.
Beginning with learners aiming at IIT JAM 2027, clarity in how equations generate such lines becomes critical. Such spectral forms fall under three main categories: emission, seen as bright marks; absorption, marked by dark bands across steady light; scattering appears apart from these two. With insight into shifts between energy levels comes access to deeper ideas – Bohr’s concept of atom structure emerges naturally, alongside the Rydberg expression, both frequently encountered in rigorous tests. Though often tested, their roots lie firmly in basic principles.
Syllabus: Physical Chemistry and Spectroscopy for IIT JAM 2027
Within the framework of IIT JAM 2027, atomic spectra appear mainly in Physical Chemistry and Spectroscopy. Beyond that domain, they emerge again within Atomic and Molecular Structure. A strong alignment exists there with IIT JAM Physical Chemistry.
Within the framework of IIT JAM 2027, atomic spectra appear mainly in Physical Chemistry and Spectroscopy. Beyond that domain, they emerge again within Atomic and Molecular Structure. A strong alignment exists there with IIT JAM coverage of Physical Chemistry.
- Core Focus: Hydrogen-like atoms, Bohr’s model, and Schrödinger wave equation applications.
- Reference Materials: Atkins’ Physical Chemistry (10th/11th ed.) and McQuarrie & Simon.
Atomic spectra For IIT JAM: An Introduction to Quantum Transitions
Atomic spectra represent the “fingerprints” of elements. For IIT JAM 2027, students must grasp that these spectra are discrete, providing direct evidence for the quantization of energy.
The Three Pillars of Spectra:
- Emission Spectra: Occur when an excited electron “drops” to a lower energy level, releasing a photon (E = hν).
- Absorption Spectra: Occur when ground-state electrons “jump” to higher levels by absorbing specific wavelengths.
- Scattering Spectra: Involve the interaction of light with the electron cloud (e.g., Raman and Rayleigh scattering).
Worked Example: Atomic spectra For IIT JAM (2027 Pattern)
Problem: A hydrogen atom in the n = 3 state decays to n = 2. Calculate the wavelength and identify the spectral series.
Solution:
- Energy Calculation: En = -13.6/n2 eV, E3 = -1.51 eV, E2 = -3.40 eV
- Energy Change: ΔE = E3 – E2 = 1.89 eV
- Wavelength (λ): hc/ΔE ≈ 1242 eV ·nm/1.89 eV = 656.3 nm
Result: This belongs to the Balmer Series (Visible region).
Misconception: Atomic spectra vs. Raman Spectroscopy
A common pitfall for IIT JAM 2027 aspirants is treating electronic transitions and Raman shifts as identical.
-
Atomic Spectra: Involve a change in the principal quantum number (n).
-
Raman Spectroscopy: Involves virtual states and changes in vibrational or rotational quantum numbers.
-
Key Tip: Atomic spectra are usually in the UV-Visible range, whereas Raman shifts are measured relative to the incident laser frequency, often in the IR-proximal region.
Application: Atomic spectra in Biotechnology
The 2027 exam landscape places heavy emphasis on interdisciplinary applications. In Biotechnology, atomic emission is used in:
-
Inductively Coupled Plasma (ICP): To detect trace metals in enzymes.
-
X-ray Photoelectron Spectroscopy (XPS): To study the surface chemistry of bio-implants.
-
DNA/Protein Labeling: Using fluorescent markers that rely on specific atomic/molecular spectral transitions.
Exam Strategy: Atomic spectra For IIT JAM 2027 Study Tips
To secure a seat in an IIT in 2027, your strategy must be data-driven.
1. Prioritize High-Yield Constants
Memorize the Rydberg constant (RH = 1.097 × 107 m-1) and the conversion factor 1 eV = 1.602 × 10-19 J.
2. Master the Selection Rules
Not all transitions are allowed. For IIT JAM 2027, remember the rule Δl = ±1. An electron cannot jump from a 2s to a 1s orbital directly via electric dipole transition.
3. Use VedPrep Resources
-
Video Lectures: Watch our 2027-specific series on “Quantum Numbers and Selection Rules.”
-
Mock Tests: Take the “Spectroscopy Mastery” quiz on the VedPrep app.
-
Notes: Access the PDF “Atomic Spectra: Beyond the Bohr Model.”
Solved Problems: Advanced Atomic spectra For IIT JAM
Q1: How many spectral lines are possible for an electron returning from n=4 to ground state?
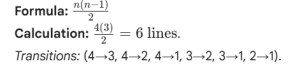
Q2: Calculate the frequency of the first line of the Lyman series for Li2+.
The Lyman series starts at n1=1, first line is n2=2. For Li2+, Z=3.
ν = c · R · Z2 (1/12 – 1/22).
Final Thoughts
When preparing for IIT JAM 2027, keep in mind that understanding Atomic Spectra goes beyond recalling equations – it builds deep insight into quantum behavior. Not merely a link between core physics and advanced chemistry, this area rewards those balancing theory with problem-solving skill. Progressing steadily – from simple Bohr atom concepts toward nuanced ideas like transition conditions and subtle energy splits – shapes strong performance. Such structured learning supports results in exams including IIT JAM and CSIR NET. Mastery emerges not from speed, but clarity gained through consistent practice.
Although mastering difficult topics takes time, consistent effort combined with official materials of VedPrep often brings measurable progress. Through carefully organized sessions paired with proven methods, improvement becomes more likely over months of steady work. One learning platform supports such growth by offering tailored tools along with experienced guidance throughout preparation phases. Clarity of thought tends to emerge when attention stays sharp across long stretches of review. Success in major assessments frequently follows those who maintain both discipline and genuine interest.
To learn more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions (FAQs)
What exactly is Atomic Spectra for IIT JAM 2027?
It is the study of discrete lines of light emitted or absorbed by atoms during electronic transitions. For the 2027 exam, it is a high-yield topic within Physical Chemistry and Quantum Mechanics.
Why are atomic spectra called "fingerprints" of elements?
Every element has a unique set of energy levels. Therefore, the wavelengths emitted or absorbed are specific to that element, allowing for precise identification.
What is the difference between emission and absorption spectra?
Emission spectra consist of bright lines on a dark background (energy released), while absorption spectra show dark lines on a continuous rainbow background (energy absorbed).
How does quantization relate to atomic spectra?
The existence of discrete spectral lines proves that electrons can only exist in specific, quantized energy states rather than a continuous range.
How do you calculate the energy of a photon in a spectral transition?
Use the relation E = hν or E = hc/λ. If you have the energy in eV, use the shortcut λ (in nm) ≈ 1242 / E (in eV).
Which spectral series falls in the visible region?
The Balmer series (nfinal = 2) is the only one visible to the human eye.
What are the Lyman and Paschen series?
The Lyman series (nfinal = 1) falls in the Ultraviolet (UV) region, while the Paschen series (nfinal = 3) falls in the Infrared (IR) region.
What is a "selection rule" in atomic spectra?
Selection rules (like Δl = ±1) determine which transitions are "allowed" or "forbidden" based on quantum mechanical probability.
How do you calculate the maximum number of spectral lines?
If an electron drops from level $n$ to the ground state, the formula is n(n-1)/2.
How does the Schrödinger wave equation apply to atomic spectra?
The equation provides the wavefunctions (ψ) and precise energy eigenvalues that define the positions of the spectral lines.
What is the Fine Structure of spectral lines?
It is the splitting of main spectral lines into closer components due to spin-orbit coupling and relativistic effects.
Is Raman Spectroscopy the same as Atomic Spectra?
No. Atomic spectra involve electronic transitions within atoms, while Raman spectroscopy involves vibrational changes in molecules through inelastic scattering.
How is atomic spectra used in Biotechnology?
It is used in techniques like ICP-OES for trace metal analysis in biological samples and in protein sequencing via absorption characteristics.
Which unit should I study for Atomic Spectra in IIT JAM 2027?
Focus on the Atomic and Molecular Structure and Spectroscopy units in the Physical Chemistry syllabus.
Are Bohr's model limitations asked in the exam?
Yes, questions often focus on why Bohr's model fails for multi-electron atoms and how quantum mechanics corrects it.













