The Maxwell-Boltzmann distribution law is one of those topics that actually makes sense once you stop looking at it as just a scary formula and start seeing it as the “demographics” of a gas. Think of it like the crowd at a busy metro station in Delhi or Mumbai. You’ve got a few people sprinting to catch a train, a few standing completely still looking at the map, and most people walking at a steady, average pace. The Maxwell-Boltzmann distribution law is just the mathematical way of describing that “crowd” of gas molecules.
Syllabus: IIT JAM Statistical Mechanics
The topic of the Maxwell-Boltzmann distribution law falls under the unit Statistical Mechanics in the IIT JAM syllabus, which is also relevant for CSIR and GATE exams. This unit is crucial in understanding the behavior of systems in thermal equilibrium.
It is covered in standard textbooks such as Problems in General Physics by I.E. Irodov, specifically in Chapter 5, and Physical Chemistry by Peter Atkins and Julio de Paula. These textbooks provide a comprehensive treatment of statistical mechanics and the Maxwell-Boltzmann distribution.
Students preparing for IIT JAM should focus on understanding the key concepts of statistical mechanics, including the Maxwell-Boltzmann distribution, which describes the distribution of speeds among gas molecules in thermal equilibrium. A thorough grasp of this topic is essential for success in the exam.
Introduction to Maxwell-Boltzmann Distribution Law For IIT JAM
At its heart, this law tells us how speeds are spread out among particles in an ideal gas. In a container of gas, molecules aren’t all moving at the same speed. They’re constantly bumping into each other, swapping energy like players in a high-stakes game of carrom.
The distribution depends on two main things: temperature and mass.
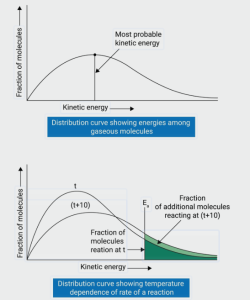
The law gives us a probability density function. If you’re looking at your Atkins or Irodov, you’ll see that the curve has a specific shape—it starts at zero, rises to a peak, and then tails off slowly toward higher speeds. This peak represents the “most probable speed.” As you crank up the heat (increase the temperature), the peak shifts to the right and gets flatter. It’s like the whole crowd starts moving faster, and the range of speeds gets wider.
Maxwell-Boltzmann Distribution Law For IIT JAM: Key Assumptions
To make the math work for exams like JAM, GATE, or CSIR NET, we have to agree on a few “rules of the game.” Here’s what we assume about our gas:
- Massive numbers: We’re talking about a huge number of particles. Statistical mechanics doesn’t really work if you’re only looking at three or four atoms.
- Total randomness: Particles move in every possible direction with no bias. There’s no “favorite” direction in the container.
- Elastic collisions: When these particles hit each other or the walls, they don’t lose energy to friction or heat. The kinetic energy just gets redistributed.
- Short-range interaction: Particles only care about each other when they actually collide. Most of the time, they’re just flying through empty space.
- Thermal equilibrium: The system has settled down. The temperature is uniform throughout, meaning the “average” behavior isn’t changing over time.
- No internal “distractions”: We assume the molecules aren’t getting excited internally (like vibrating or rotating differently) during these collisions. We’re strictly focused on how fast they’re moving from point A to point B—their translational kinetic energy.
Worked Example: Maxwell-Boltzmann Distribution Law For IIT JAM
The Maxwell-Boltzmann distribution law describes the distribution of speeds among gas molecules at thermal equilibrium. A sample of oxygen molecules (O2) is maintained at a temperature of 300 K. Calculate the average speed and the most probable speed of the oxygen molecules.
The average speed <v> and the most probable speed vmp can be calculated using the following equations:
- <v> =(8RT/πM)¹/²
- vmp = (2RT/M)¹/²
where R is the gas constant (8.314 J/mol·K), T is the temperature in Kelvin, and M is the molar mass of the gas. For oxygen, M = 32 g/mol = 0.032 kg/mol.
| Speed | Equation | Calculation | Result (m/s) |
| Average speed | <v> = (8RT/πM)¹/² | (8 × 8.314 × 300 / (π × 0.032)) 1/2 | 478 |
| Most probable speed | vmp = (2RT/M)¹/² | (2 × 8.314 × 300 / 0.032)¹/² | 395 |
The Maxwell-Boltzmann distribution law For IIT JAM often involves such calculations, which help in understanding the behavior of gases. These speeds are essential in determining various properties of gases.
Misconceptions in Understanding Maxwell-Boltzmann Distribution Law For IIT JAM
A big trap many students fall into is thinking that the peak of the curve represents the average speed of the molecules. In reality, that highest point is just the “most probable speed”—the speed most particles happen to have. If you’re stuck in a Bangalore traffic jam, most cars might be crawling at 10 km/h, but the average speed of everyone on the road is actually higher because of a few clear stretches. Similarly, the Maxwell-Boltzmann distribution law curve isn’t symmetrical like a standard bell curve; it has a long tail toward higher speeds, which pulls the average and RMS speeds to the right of the peak.
Another common slip-up is forgetting how temperature changes the graph. People often think heating a gas just shifts the curve, but it actually flattens it out too. You aren’t just moving the peak; you’re spreading the energy across a wider range of possibilities. Also, don’t confuse this with the behavior of quantum particles—this law only works when your particles are “distinguishable” and moving classically.
Application of Maxwell-Boltzmann Distribution Law For IIT JAM
The Maxwell-Boltzmann distribution law has numerous real-world applications, particularly in understanding the behavior of gases. One significant application is in the field of chemical engineering, where it is used to model and predict the behavior of gases in various industrial processes. This law helps engineers design and optimize systems involving gas mixtures, such as gas separation and purification processes.
When you’re solving problems for JAM, you’ll see the Maxwell-Boltzmann distribution law popping up everywhere, not just in theory. Its biggest job is helping us calculate things we can actually measure, like the pressure a gas exerts on its container. Since pressure comes from particles hitting the walls, knowing their speed distribution is the only way to get an accurate reading.
It’s also the secret behind why chemical reactions happen faster when you turn up the heat. In a reaction, only the “high-energy” particles—the ones in that long tail of the curve—have enough speed to break bonds. By using this law, you can figure out exactly what fraction of molecules has enough kick to get the job done. In the lab, this helps explain everything from how stars burn to how a simple pressure cooker works. For your exam, mastering these applications helps you connect abstract statistical math to real-world thermodynamics.
Exam Strategy for Maxwell-Boltzmann Distribution Law For IIT JAM
When you’re staring at the paper on exam day, the trick to mastering the Maxwell-Boltzmann distribution law is all about spotting ratios and shifts. Don’t just memorize the formulas for RMS, average, and most probable speeds; remember their order like a mental shortcut. Since the RMS is always the largest and the probability is the smallest, you can quickly eliminate wrong options in multiple-choice questions (MCQs) just by looking at the values.
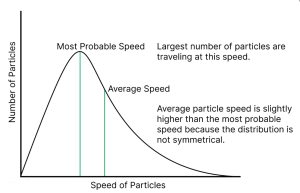
Pay close attention to how the curve changes when the mass of the gas or the temperature shifts. If the question gives you a heavier gas like Argon versus a lighter one like Helium, visualize that curve stretching or squeezing. For those tricky Numerical Answer Type (NAT) questions, keep your units consistent—sticking to SI units is usually the safest bet to avoid silly calculation errors. If you can sketch the graph and mark the key points in your head, you’ll find these questions much easier to handle.
Students should concentrate on solving problems related to the Maxwell-Boltzmann distribution, such as finding the most probable speed, average speed, and root mean square speed of gas molecules. Practice is key to mastering these problems. VedPrep provides expert guidance and practice materials to help students build a strong foundation in statistical mechanics.
VedPrep offers a range of resources, including practice questions, video lectures, and online tests, to help students prepare for IIT JAM. By following a structured study plan and utilizing VedPrep’s resources, students can develop a deep understanding of the Maxwell-Boltzmann distribution law and improve their problem-solving skills.
Key Equations and Formulas for Maxwell-Boltzmann Distribution Law For IIT JAM
1. The Probability Density Function
The Maxwell-Boltzmann distribution P(v) describes the probability of finding a particle with speed v at a given temperature T.
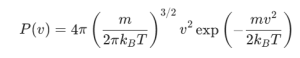
-
m: Mass of a single molecule
-
kB: Boltzmann constant (1.38 × 10-23 J/K)
-
T: Absolute temperature (in Kelvin)
2. Energy Distribution Formula
Sometimes the exam targets the kinetic energy (E) rather than speed (v). The distribution for kinetic energy in 3D is:
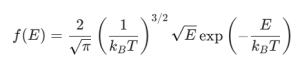
Final Thoughts
Wrapping things up, the Maxwell-Boltzmann distribution law isn’t just another page of math to flip through; it’s basically the heartbeat of how we understand gases. When you finally get how temperature and mass dance together to reshape that curve, the rest of thermodynamics starts falling into place like pieces of a puzzle. It’s like finally understanding the flow of a crowd at a local mela—it looks like chaos at first, but there’s a very specific logic behind it.
As you keep pushing through your prep, don’t let the complex-looking integrals throw you off. Focus on the physical meaning behind the peaks and tails of the graph. If you can explain the concept to a friend over a quick chai break, you’re definitely on the right track. Keep grinding through those previous year papers, stay consistent, and you’ll find yourself clearing these hurdles with confidence.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions (FAQs)
Why is the Maxwell-Boltzmann distribution important for IIT JAM?
It is a core topic in both Physical Chemistry and Physics syllabi. It forms the foundation for understanding kinetic theory, reaction rates, and statistical mechanics.
How does increasing temperature affect the distribution curve?
As temperature increases, the peak of the curve shifts to the right (higher speeds) and the peak height decreases as the distribution flattens or "broadens."
How does the mass of a gas molecule affect its speed distribution?
Heavier molecules move more slowly on average. Therefore, the curve for a heavier gas (like O2) is narrower and taller compared to a lighter gas (like H2) at the same temperature.
Does the Maxwell-Boltzmann distribution apply to liquids?
Technically, it is derived for ideal gases. While similar statistical models apply to liquids, the standard Maxwell-Boltzmann equations assume negligible intermolecular forces, which isn't true for liquids.
What happens to the fraction of molecules with high energy as temperature rises?
The fraction of molecules with high kinetic energy increases significantly. This is why chemical reactions occur faster at higher temperatures.
Is the Maxwell-Boltzmann distribution a quantum or classical model?
It is a classical statistical model. It assumes particles are distinguishable and does not account for quantum effects like the Pauli Exclusion Principle.
What is the area under the Maxwell-Boltzmann curve represent?
The total area under the probability density curve is always equal to 1 (or 100%), representing the total probability of finding a molecule at any speed.
What are the most common mistakes in MB Distribution problems?
The most common errors are forgetting to convert Celsius to Kelvin and failing to convert molar mass from grams to kilograms.
Are numerical questions on this topic frequent in IIT JAM?
Yes, usually 1-2 questions appear annually, often involving ratio calculations between different gases or temperatures.
Do I need to memorize the long probability density function?
While the derived speed formulas are more important for numericals, understanding the v2 and exponential terms of the function is necessary for conceptual/graphical questions.
How is this topic related to the Collision Theory of reaction rates?
Collision theory uses this distribution to calculate the number of molecules that possess enough energy (Activation Energy) to react upon collision.
Can I use this law for real gases?
It works well for real gases at high temperatures and low pressures where they behave ideally. At very high pressures, deviations occur.
Where can I find practice problems for IIT JAM 2027?
VedPrep provides specialized modules and previous year question (PYQ) analysis specifically for Statistical Mechanics and Kinetic Theory.














