The Freundlich adsorption isotherm is a mathematical model describing the adsorption of gases onto solid surfaces, widely used in IIT JAM and CSIR NET chemistry.
Freundlich adsorption isotherm For IIT JAM
If you’re diving into physical chemistry for IIT JAM, you already know that surface chemistry is one of those high-yield areas where you can score some relatively easy marks if your basics are solid. Within that, the Freundlich adsorption isotherm is a total staple. It shows up in the thermodynamics and kinetics section of the JAM syllabus, and honestly, if you’re also eyeing CSIR NET or GATE down the road, you’re going to see this concept over and over again.
To really get under the hood of this topic, most of us end up leaning on the classics. Atkins is great for the heavy lifting on principles, and Levine is a lifesaver when you need a deep dive into the math. At VedPrep, we’ve noticed that students who master this early have a much smoother ride when they hit the more complex industrial applications later on.
For in-depth study, students can refer to standard textbooks such as the following:
- Atkins, Physical Chemistry, 10th Edition—a comprehensive resource that covers the principles of physical chemistry, including adsorption isotherms.
- Levine, Physical Chemistry, 6th Edition—another widely used textbook that provides detailed explanations of surface chemistry and adsorption phenomena.
Freundlich Adsorption Isotherm: Definition and Mathematical Expression
Let’s keep it simple. An adsorption isotherm is basically just a graph. It shows how much gas or liquid gets “stuck” to a solid surface as you change the pressure, all while keeping the temperature steady. The Freundlich adsorption isotherm is an empirical model—which is just a fancy way of saying it’s based on what scientists actually saw happening in the lab rather than just a pure theory.
The math looks like this:
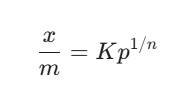
Here’s the breakdown of what those letters actually mean:
-
x: The amount of stuff (adsorbate) that’s stuck.
-
m: The mass of the solid (adsorbent) doing the sticking.
-
P: The pressure of the gas.
-
K and n: These are constants. They change depending on the materials you’re using.
Basically, as you crank up the pressure, more gas molecules get forced onto the surface, so x/m goes up. Think of it like a sponge—the harder you press a damp sponge against a spill, the more it’s going to soak up until it hits a limit.
Freundlich Adsorption Isotherm For IIT JAM: Key Features and Assumptions
The big thing to remember for JAM is that Freundlich assumes a heterogeneous surface. Imagine a rocky mountain face instead of a smooth glass table. Some spots on that mountain are perfect for a hiker to grab onto (high energy sites), while others are slippery and useless (low energy sites).
Key things to keep in mind:
-
It’s not ideal: It doesn’t pretend every spot on the surface is the same.
-
Heterogeneous surface: Adsorption happens on different types of sites with different energies.
-
Finite capacity: It acknowledges that eventually, you can’t just keep adding more and more stuff.
While models like the Langmuir isotherm assume everything is perfect and uniform, the Freundlich adsorption isotherm is more like the “real world” version that accounts for all the bumps and imperfections on a surface.
Freundlich Adsorption Isotherm: Worked Example For CSIR NET
Imagine you’re given a set of data where the amount of gas adsorbed triples when the pressure is increased by a factor of nine. You can use the log version of the formula to find n:
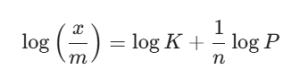
By plotting x/m against log P, you get a straight line. The slope of that line is your 1/n. If you’re stuck on these types of calculations, don’t sweat it—we see JAM aspirants trip up on the log math all the time at VedPrep, but it’s just a bit of practice.
Misconceptions in Freundlich Adsorption Isotherm For IIT JAM
A huge mistake students make is thinking this is an “ideal” model. It’s definitely not. Ideal models assume the surface is as flat and predictable as a fresh sheet of ice. But the Freundlich adsorption isotherm knows better—it assumes the surface is messy and varied (heterogeneous).
Because the surface sites have different “clinginess” (energies), the molecules don’t spread out perfectly. This is why the equation doesn’t show a simple 1:1 direct proportionality. It’s a bit more nuanced than that, which makes it way more accurate for most actual chemistry experiments you’ll run into.
Application of Freundlich Adsorption Isotherm in Real-World Scenarios
Why do we care about this? Because it’s how we clean up the planet.
-
Cleaning Water: If you’ve ever used a charcoal water filter, you’ve used this principle. The activated carbon pulls out impurities from the water based on these isotherms.
-
Industrial Cleanup: Factories use this to design systems that scrub toxic gases out of their exhaust before it hits the atmosphere.
-
Catalysis: It helps chemists understand how reactants sit on a catalyst to make a reaction go faster.
A quick (fictional) example: Imagine a small town’s water supply gets contaminated with a specific dye from a nearby factory. Engineers don’t just guess how much carbon they need to clean it. They use the Freundlich adsorption isotherm to calculate exactly how much adsorbent (the carbon) is needed to “trap” the $x$ amount of dye molecules at a certain concentration. Without this math, they’d either waste money on too much carbon or, worse, leave the water messy.
Exam Strategy for Freundlich Adsorption Isotherm For IIT JAM
When you’re sitting in that exam hall, the questions usually boil down to three things: the equation, the graph, or the limitations.
-
Memorize the log form: It’s almost always easier to solve problems using log(x/m) = log K + 1/n log P.
-
Know the constants: Understand what happens if n = 1 or if n is very large.
-
Practice the graphs: Being able to look at a curve and know if it’s Freundlich or Langmuir is a superpower.
At VedPrep, we usually suggest our students practice at least 10-15 different variations of these problems. Once you see the pattern, it’s hard to forget.
Key Limitations and Criticisms of Freundlich Adsorption Isotherm
As helpful as it is, this model isn’t perfect.
-
No Surface Diffusion: It ignores the fact that molecules might slide around on the surface after they land.
-
Low Pressure Failure: If the pressure is super low, the math starts to fall apart and doesn’t match what we see in real life.
-
Temperature Issues: It’s mostly an “at one temperature” deal. If you start heating things up, the Freundlich model doesn’t stay very accurate.
-
No Real Theory: Since it’s empirical (based on observation), it doesn’t really explain why the molecules are sticking—it just tells you that they are.
Conclusion
The Freundlich adsorption isotherm is one of those concepts that might look a bit intimidating on paper with its logs and exponents, but it’s actually pretty grounded in how the real world works. For any IIT JAM aspirant, mastering this isn’t just about ticking off a box in the syllabus—it’s about understanding the “messy” side of chemistry where surfaces aren’t perfect and reactions don’t always follow the ideal rules.
To know more in detail from our expert faculty, watch our YouTube Video:
Frequently Asked Questions
What is the standard mathematical formula for it?
The formula is x/m = Kp1/n, where $x/m$ is the extent of adsorption, P is pressure, and K and n are constants.
What does "empirical" mean in this context?
It means the model is based on experimental observations and data rather than being derived from a theoretical first principle.
What is the physical significance of x and m?
x represents the mass of the adsorbate (the gas), and m is the mass of the adsorbent (the solid surface).
What does the slope of the Freundlich plot represent?
In a plot of log(x/m) vs log P, the slope is equal to 1/n.
What does the intercept represent in the log plot?
The intercept on the y-axis gives you the value of log K.
Is this isotherm used for physical or chemical adsorption?
While it can apply to both, it is most commonly used to describe physical adsorption (physisorption) on rough surfaces.
What is an "isotherm" anyway?
It’s a curve that shows the relationship between two variables (like pressure and adsorption) while the temperature stays exactly the same.
Does this isotherm work at very high pressures?
No. A major mistake is applying it to high-pressure scenarios where adsorption usually becomes independent of pressure (reaches saturation).
Do I need to worry about units?
Yes. Always check if the pressure is in atm, bar, or torr, as this will change the value of K.
What happens to the equation at very low pressure?
At very low pressure, $1/n$ often approaches 1, meaning adsorption is directly proportional to pressure (x/m ∝ P1).
How does temperature affect the constants K and n?
Since adsorption is generally exothermic, as temperature increases, the extent of adsorption decreases, which changes these constant values.
What is a "heterogeneous surface" in Freundlich theory?
It means the surface has sites with different binding energies. Some spots are "stickier" than others.
How does it differ from the Langmuir Isotherm?
Langmuir assumes a perfectly flat, uniform surface where every spot is the same. Freundlich assumes the surface is bumpy and varied.
What is the significance of the value of n?
It’s a measure of the intensity of adsorption. Usually, if n > 1, it indicates favorable adsorption.














