Real gases and van der Waals equation of state is a critical topic in IIT JAM physics, involving corrections to the ideal gas equation to account for intermolecular forces. It’s essential to understand the concept, derivation, and applications for a strong score in the exam.
Syllabus: Thermodynamics and Statistical Mechanics (IIT JAM Physics Syllabus)
Real gases and van der Waals equation of state fall under thermodynamics and statistical mechanics of the official IIT JAM syllabus. The Thermodynamics and Statistical Mechanics unit covers essential concepts, including equations of state for real gases.
Key textbooks that cover this topic include:
- Halliday, Resnick and Walker- This widely used textbook provides an in-depth explanation of thermodynamic properties and the behavior of real gases.
- Atkins and De Paula- This popular physical chemistry textbook also covers the van der Waals equation and its significance in understanding real gas behavior.
Students preparing for the IIT JAM Chemistry exam should focus on understanding the van der Waals equation, its derivation, and applications. A thorough grasp of thermodynamic concepts, including equations of state, is required for success in the exam.
Real Gases and van der Waals Equation of State: Concept and Derivation
The ideal gas equation, PV = nRT, is like that perfect study schedule we all make—it looks great on paper but rarely survives contact with reality. As per Real gases and van der Waals equation of state, it assumes gas molecules are tiny points with zero volume and absolutely no interest in talking to each other. But in the real world, gases are a bit more complicated. Real gases deviate from that “perfect” behavior because, let’s face it, molecules do take up space and they definitely feel the pull of their neighbors.
To fix this, the van der Waals equation of state steps in. Instead of pretending everything is ideal, it adds “corrections” to the math. Think of it as adjusting your study plan to account for the time you actually spend on coffee breaks. It introduces two constants, a and b, to handle intermolecular forces and the physical size of the molecules.
The equation looks like this:
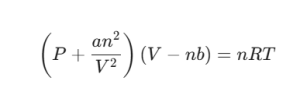
Here, a and b are the “personality traits” of the gas. The term an2/V2 fixes the pressure because molecules attract each other, slowing down their impact against the container walls. Meanwhile, b accounts for the “excluded volume”—the space the molecules themselves occupy that nothing else can enter.
Real gases and van der Waals equation of state For IIT JAM
A huge mistake many of us make while prepping for the IIT JAM is thinking that “non-ideal” just means the molecules are big. That’s only half the story. The real secret sauce of the Real gases and van der Waals equation of state is understanding those intermolecular forces.
Based on a scenario of Real gases and van der Waals equation of state, imagine you’re at a crowded concert. If everyone is far apart, you can move wherever you want (that’s low pressure/high temp). But as the crowd gets packed, you start bumping into people (molecular volume), and maybe you’re trying to move toward the stage, but your friends are pulling you back for a selfie (attractive forces).
These interactions become a big deal when you crank up the pressure or drop the temperature. For exams like IIT JAM, you need to know that these forces are what keep the gas from being “ideal” in Real gases and van der Waals equation of state. At VedPrep, we often see students struggle with the physical meaning of a and b. Just remember: a is about the “stickiness” (attraction), and b is about the “bulkiness” (size).
Real gases and van der Waals equation of state For IIT JAM
Why does this matter outside of a textbook? Well, if you’re designing a chemical reactor or working in a lab, using PV=nRT might actually be dangerous because it under-predicts how much pressure a gas will exert under certain conditions.
-
Engineering: Designing tanks for gases like CO2 or CH4 requires knowing exactly when they’ll liquefy.
-
Petroleum: In deep underground reservoirs, the pressures are so high that the ideal gas law basically gives up. Engineers use van der Waals (or even more complex versions) to keep things running safely.
| Gas | Critical Temperature (K) | Critical Pressure (bar) |
|---|---|---|
| CO2 | 304.2 | 73.9 |
| CH4 | 190.6 | 45.9 |
If you’re aiming for a top rank, you’ll want to get comfortable with these values. They tell us the limit where a gas can no longer be forced into a liquid state, no matter how much pressure you apply.
Real gases and van der Waals equation of state For IIT JAM
Cracking the Real gases and van der Waals equation of state part of the IIT JAM syllabus requires a bit of a game plan. You can’t just dive into the hard math; you need to see the “why” behind it.
What to focus on:
-
The “Why”: Why does the ideal law fail? (Hint: High P, Low T).
-
The Constants: What do the units of a and b actually mean?
-
Isotherms: Learn to read those P-V curves. They show you exactly where the gas starts acting weird.
A good way to study is to start with the basics of PV=nRT and then “break” it. Try to see how much the volume changes when you add that nb correction. VedPrep has a lot of practice sets that walk you through these specific types of problems so you aren’t caught off guard by a tricky question about critical constants.
VedPrep’s resources are tailored to meet the needs of CSIR NET, IIT JAM, and GATE aspirants, providing a comprehensive platform for exam preparation.
Derivation of van der Waals Equation of State: Step-by-Step Solution
The derivation is actually pretty logical when you break it down into two steps. We start with the ideal version and apply “reality checks.”
-
Volume Correction: In an ideal gas, the “available” volume is the whole container (V). But real molecules take up space. So, the space actually available for movement is V – nb.
-
Pressure Correction: In a real gas, a molecule headed for the wall gets “pulled back” by the molecules behind it. This reduces the force of the impact. To find the “ideal” pressure, we have to add back the pressure lost to this attraction: (P + an2/V2)
When you put those two corrected terms together into the original PV=nRT format, you get the full van der Waals equation under Real gases and van der Waals equation of state. It’s a much more accurate way to look at the world, especially when things get tight and cold.
Limitations and Approximations of van der Waals Equation of State
Even though it’s a massive upgrade, the van der Waals model isn’t perfect. It’s a “mean-field” theory, which is a fancy way of saying it averages things out.
The biggest red flag? It predicts these weird “loops” in the P-V graph during the transition from gas to liquid that don’t actually happen in real life. It also treats molecules like hard spheres. If you have a long, floppy molecule or something with a lot of charge (like water vapor), this equation starts to lose its edge. For those cases, you’d need even more advanced models like Peng-Robinson, but for the IIT JAM, mastering van der Waals is your main priority.
Final Thoughts
Getting a handle on the Real gases and van der Waals equation of state is really about moving from “perfect world” physics to “real world” physics. It’s one of those topics that ties together everything from molecular behavior to industrial engineering.
As you gear up for the IIT JAM, don’t just treat a and b as boring numbers while covering Real gases and van der Waals equation of state. Think of them as the physical traits that make each gas unique. Keep practicing those isotherms and critical point calculations. At VedPrep, we’ve seen that the students who really “visualize” the molecules bumping into each other are the ones who sail through the thermodynamics section.
To learn more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
How does the van der Waals equation differ from the ideal gas law?
The van der Waals equation introduces two correction terms: a/V2 for intermolecular attractions and b for the finite volume of gas molecules, making it more accurate for real-world conditions.
What are the units of the van der Waals constants 'a' and 'b'?
The constant a is typically measured in L2 atm mol-2, while b is measured in L mol-1.
What does the 'a' constant represent?
The constant a represents the magnitude of the attractive forces between the gas molecules. A higher a value indicates stronger intermolecular attractions.
What does the 'b' constant represent?
The constant b represents the "excluded volume" or the actual volume occupied by the gas molecules themselves per mole.
Why do real gases deviate from ideal behavior at high pressure?
At high pressure, the volume of the container decreases, making the space occupied by the molecules themselves significant. Additionally, molecules are closer together, increasing intermolecular forces.
Under what conditions does a real gas behave most like an ideal gas?
Real gases behave most ideally at low pressure and high temperature, where intermolecular forces are negligible and the molecular volume is tiny compared to the total volume.
What is the "excluded volume"?
The excluded volume is the volume around each molecule that is unavailable for other molecules to occupy. It is approximately four times the actual molecular volume.
Can the van der Waals equation predict phase changes?
It qualitatively describes the transition between gas and liquid, but it often predicts non-physical "loops" (van der Waals loops) that require the Maxwell construction to fix.
What is the formula for the van der Waals equation for 'n' moles?
The formula is: (P + an2/V2)(V - nb) = nRT.
Which gas would have a higher 'a' value: Helium or Ammonia?
Ammonia (NH3) has a much higher $a$ value because it is a polar molecule with strong intermolecular forces, whereas Helium has very weak London dispersion forces.
Is the van der Waals equation applicable to liquids?
While it can provide insights into liquid behavior, it is primarily designed for gases and becomes significantly less accurate in the liquid phase.
What is the Boyle temperature?
The Boyle temperature is the specific temperature at which a real gas behaves most ideally over a range of pressures because the attractive and repulsive forces cancel each other out.
Why does the ideal gas law underestimate the pressure of a real gas at very high pressures?
At very high pressures, the volume of the molecules (repulsive effect) dominates, making the gas harder to compress and resulting in a higher pressure than predicted by PV=nRT.
What is the main limitation of this equation?
It fails to accurately predict the behavior of gases at the critical point and does not account for the temperature dependence of a and b.














