The Langmuir adsorption isotherm is a mathematical model used to describe the adsorption of molecules onto a surface. It is a fundamental concept in physical chemistry and is often tested in IIT JAM and CSIR NET exams.
Understanding Langmuir Adsorption Isotherm: Syllabus and Key Textbooks
If you’re gearing up for the IIT JAM, you’ve probably realized that Surface Chemistry is one of those units where you can really score well if you get the basics right. The Langmuir adsorption isotherm is a superstar in this section. It’s not just a fancy equation; it’s a fundamental way to describe how gas molecules “stick” to a solid surface.
At VedPrep, we’ve noticed that students often get overwhelmed by the math, but it’s actually pretty straightforward once you see the logic behind it while covering areas such as Langmuir adsorption isotherm. You’ll find this topic under Physical Chemistry in the official syllabus. If you want to dive deeper, Atkins’ Physical Chemistry is the classic go-to. For those who want to get their hands dirty with practice problems, Irodov’s General Physics has some great challenges, though it can be a bit intense.
Langmuir Adsorption Isotherm For IIT JAM: Main Concept Explanation
Imagine you’re at a busy coffee shop (like the one we’re virtually sitting in) and there are only ten bar stools. Once someone sits on a stool, that spot is taken. No one can sit on top of them, and nobody is fighting over the same seat. This is basically what the Langmuir adsorption isotherm describes—but with molecules and surfaces.
The equation looks like this:
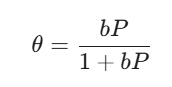
Here, θ is the “surface coverage” (think of it as the percentage of bar stools occupied), P is the pressure of the gas, and b is a constant that tells us how strongly the gas wants to stick to the surface.
This model relies on a few big “what ifs” (assumptions):
A Flat Playground: The surface is perfectly smooth and uniform (homogeneous). Every “seat” is identical.
One Layer Only: Once a molecule grabs a spot, no other molecule can pile on top of it. It’s a monolayer.
Mind Your Own Business: The molecules don’t interact with their neighbors. They just sit there and mind their own business.
Of course, real life is messier. Real surfaces have “potholes” (defects), and sometimes molecules like to pile up in layers. That’s where this model falls a bit short, but for the JAM, mastering these basics is your first step to success.
Worked Example
Let’s look at a quick calculation. Suppose a gas is hitting a surface at 1 atm pressure. If our constant b is 0.1, what’s the coverage?
Using the formula:
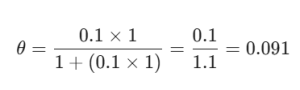
So, about 9.1% of our surface is covered. It’s like having a 100-seat lecture hall and only 9 students showing up—plenty of room left! At (Company Name), we suggest playing around with these numbers to see how increasing the pressure P eventually fills up the surface.
Common Misconceptions About Langmuir Adsorption Isotherm For IIT JAM
A big trap students fall into is thinking the surface can be “bumpy” or varied (heterogeneous). In Langmuir’s world, every single spot on that surface is exactly the same. If you start talking about different energy levels on the same surface, you’ve moved out of Langmuir territory.
Another common slip-up is forgetting that this is monolayer only. If the question mentions molecules stacking like a skyscraper, Langmuir isn’t your guy. Remembering these small details can be the difference between a “plus mark” and a “minus mark” on your exam paper.
Real-World Applications
This isn’t just for passing exams; this stuff is everywhere.
Cleaning Water: Think of activated carbon in a water filter. It’s like a giant sponge for pollutants. The dye molecules stick to the carbon surface just like the gas molecules in our model.
Gas Separation: Zeolites act like high-tech filters that grab specific gases while letting others pass by.
Health Tech: Many biosensors work by having specific proteins stick to a surface to trigger a reading.
At VedPrep, we find that thinking about these real-world uses makes the “dry” equations a lot more interesting to study.
Exam Strategy for Langmuir Adsorption Isotherm For IIT JAM
When you’re sitting in that exam hall, don’t just jump into the math. Read the question carefully to see if the assumptions hold up. If the problem mentions “low pressure,” remember that the equation simplifies to θ ≈ bP. If it’s “high pressure,” the surface is saturated, and θ stays close to 1.
Practice is everything. Try to solve for b or P by rearranging the formula. We recommend looking at previous years’ papers to see how they twist these questions. It’s usually about finding one missing variable while keeping the assumptions in mind.
Comparison of Langmuir Adsorption Isotherm with Other Adsorption Isotherms
Not all isotherms are created equal. Here’s a quick breakdown:
| Isotherm | Surface Type | Layers | Best Used For |
| Langmuir | Homogeneous | Monolayer | Basic gas-solid adsorption |
| Freundlich | Heterogeneous | Multilayer (can be) | Complex, “rough” surfaces |
| BET | Homogeneous | Multilayer | Finding the total surface area |
Practice Problems – Langmuir Adsorption Isotherm For IIT JAM
Let’s try one more to keep your brain sharp.
Suppose you have a gas where b = 0.5 and the pressure is 2 atm.
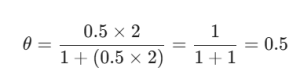
In this case, the surface is exactly half-full. If you can do that, you’re already on the right track! Keeping things simple and focusing on the core logic is how we at (Company Name) like to approach these topics. Good luck with your prep—you’ve got this!
Final Thoughts
The Langmuir adsorption isotherm isn’t just a scary-looking equation to memorize for the IIT JAM; it’s a logical way to look at how molecules find a home on a surface. Whether you’re thinking about how a water filter cleans your drink or how a biosensor detects a virus, the core idea is the same: once a spot is taken, it’s taken. At VedPrep, we know that exam prep can feel like a marathon, but breaking down these physical chemistry concepts into relatable scenarios makes the finish line feel a lot closer. Keep practicing those numericals, stay clear on your assumptions, and you’ll be well on your way to acing the surface chemistry section.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
Why is it called an "isotherm"?
"Iso" means same, and "therm" means temperature. We call it an isotherm because the whole process is studied while keeping the temperature constant. If you change the temperature, the equilibrium—and the graph—shifts.
What are the three big assumptions I need to remember for IIT JAM?
Just remember: homogeneous surface (all spots are equal), monolayer (no stacking), and no lateral interactions (molecules don't talk to or push their neighbors).
What does the "b" in the equation represent?
The constant b is essentially the "stickiness" factor. It’s the ratio of the rate of adsorption to the rate of desorption. A higher b value means the gas really likes staying on the surface.
Does Langmuir account for "physical" or "chemical" adsorption?
While it can be used for both, its assumption of a single monolayer makes it a much better fit for chemisorption, where molecules form strong chemical bonds with the surface.
Why doesn't Langmuir work at very high pressures in real life?
In reality, once the first layer is full, gas molecules often start piling on top of each other. Langmuir’s model says "no stacking allowed," so it fails when multilayer adsorption starts happening.
What happens to the equation at very low pressure?
At low pressure, the bP in the denominator becomes tiny compared to 1. The equation simplifies to θ= bP. This means the coverage is directly proportional to the pressure.
Can I use Langmuir for liquids?
Yes. You can adapt it for solutes in a solution. Instead of pressure (P), you just use the concentration (C) of the substance in the liquid.
What is "surface coverage" (θ)?
It’s just a fraction. If θ is 0.7, it means 70% of the available spots on the adsorbent are occupied by gas molecules.
Does the Langmuir model consider the size of the molecules?
Not directly in the basic equation. It assumes each molecule takes up exactly one site, regardless of how big or small the molecule actually is.
What's a real-life example of a "homogeneous" surface?
Perfectly smooth single crystals in a lab are close. However, most things we use at VedPrep, like charcoal or clay, are actually quite "bumpy" and heterogeneous.
Is adsorption exothermic or endothermic according to this model?
Adsorption is generally exothermic (releases heat). According to Le Chatelier’s principle, if you crank up the heat, the amount of gas adsorbed will actually go down.
What is the "saturation" point?
That's the moment when every single available site on the surface is occupied. No matter how much more pressure you add, you can't stick any more molecules onto the surface.
How often does this show up in IIT JAM?
Almost every year! Whether it's a direct numerical or a conceptual question about the assumptions, it’s a high-yield topic you shouldn't skip.
How do I calculate the volume of gas adsorbed at STP using Langmuir?
You’d typically use the volume-based version of the formula: V = (Vm · bP) / (1 + bP), where Vm is the volume of gas needed to form a complete monolayer.














