Equipartition of energy is a fundamental concept in thermodynamics that states that in a closed system, the total energy is equally distributed among its degrees of freedom, leading to an equilibrium state. Understanding this concept is essential for IIT JAM and other competitive exams.
Equipartition of Energy For IIT JAM: Understanding the Concept
Let’s talk about something that sounds heavy but is actually pretty intuitive once you peel back the layers: the equipartition of energy. If you’re prepping for the IIT JAM, you know thermodynamics is a huge chunk of the syllabus. This concept is basically the “fairness doctrine” of the molecular world.
At its heart, the theorem says that if a system is in thermal equilibrium, the total energy doesn’t just hang out in one place. Instead, it gets split up equally among all the different ways a molecule can move. We call these “ways to move” degrees of freedom.
Think of it like a group of friends ordering a giant pizza. If everyone is equally hungry and the group is in “equilibrium,” each person (or degree of freedom) gets an equal number of slices. In the world of physics, each “slice” of energy is exactly ½ kT.
Key Characteristics:
-
Every single degree of freedom grabs ½ kT of energy (k is the Boltzmann constant, T is temperature).
-
The energy is spread out like a perfectly divided bill at dinner.
-
This only works when things have settled down into thermal equilibrium.
Syllabus – Thermodynamics
At VedPrep, we’ve noticed that students often breeze past this topic, thinking it’s just one formula. But in the IIT JAM syllabus section, it’s the backbone of understanding heat capacities (Cp and Cv).
You’ll find this in the classic “holy grail” books like Atkins to cover areas such as equipartition of energy. The core idea you need to grasp for the exam is how internal energy (U) connects to these degrees of freedom. It’s the bridge between the tiny world of atoms and the big world of temperature readings.
Equipartition of Energy For IIT JAM: Key Formulas and Equations
Alright, let’s get into the math, but we’ll keep it simple. If a system has $n$ degrees of freedom, the total energy E is:
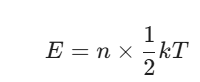
If you’re looking at a single degree of freedom (let’s call it εi), its average energy is:
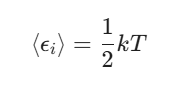
Let’s break down the degrees of freedom (DOF):
-
Monatomic Gas (like Helium): It’s just a tiny dot. It can move up/down, left/right, and forward/backward. That’s 3 translational DOFs. Total energy = 3/2 kT.
-
Diatomic Gas (like O2): Imagine a dumbbell. It can move in 3 directions, but it can also rotate like a spinning baton. That adds 2 rotational DOFs. Total energy = 5/2 kT.
Worked Example: Equipartition of Energy for IIT JAM
Imagine you have a container filled with N = 1020 molecules of a specific gas. Let’s say this gas is diatomic, so it has 5 degrees of freedom (3 translational + 2 rotational). If the room is at a comfortable 300 K, how much energy are we talking about?
-
Identify the bits: f = 5, N = 1020, T = 300 K, and k = 1.38 × 10-23 J/K.
-
Plug it in: E = f/2 NkT
-
Do the math: E = 5/2 × 1020 × 1.38 × 10-23 × 300
-
Result: You get about 5.175 Joules.
It doesn’t sound like much, but for 1020 tiny molecules, that’s a lot of microscopic dancing!
Misconception: Equipartition of Energy vs. Conservation of Energy
This is a classic trap. We’ve seen plenty of smart students at VedPrep get these two mixed up.
Conservation of energy is the old “energy can’t be created or destroyed” rule. It’s about the total amount staying the same. Equipartition of energy is about the distribution.
The Coffee Shop Scenario: > Imagine a fictional scenario where you and four friends go to a coffee shop. Conservation of energy says that if the group brings $50, the group still has $50 after they walk inside. Equipartition says that once you sit down to order, you’ll likely decide to split that $50 so everyone has exactly $10 to spend. One is about the total; the other is about the split.
Equipartition tells us how the energy likes to “settle” once the system finds its groove, or equilibrium.
Exam Strategy: Tips for Solving Equipartition of Energy Questions
When you’re staring down a JAM paper, the questions usually hide the degrees of freedom. They won’t always tell you “this molecule has 5 DOFs.” They’ll say, “Nitrogen at room temperature. ” You have to know that nitrogen is diatomic and, at room temperature, its vibrations haven’t “woken up” yet.
Quick Tips:
-
Check the Temperature: Vibrational degrees of freedom usually only count at very high temperatures. For most JAM problems, stick to translation and rotation unless the question says otherwise.
-
now your Cv: Remember that Cv = (∂U/∂T)v. Since U = f/2 RT (for one mole), Cv is just f/2 R.
-
Practice: We always tell our students that seeing the formula isn’t the same as using it. Try applying it to different gases like CO2 (linear) vs H2O (non-linear).
VedPrep provides students with access to practice problems and solutions, allowing them to assess their knowledge and identify areas for improvement. By following these tips and practicing consistently, students can develop a strong grasp of equipartition of energy and excel in their exams.
Solved Problems: Equipartition of Energy For IIT JAM
Let’s look at something a bit more advanced—spins in a magnetic field.
If you have a bunch of non-interacting “spins” (think of them as tiny magnets), they can either point up or down. In a magnetic field, these two states have different energies (+μB$ and -μB).
While the math involves something called a partition function (Z), the big takeaway is how the energy behaves at high temperatures. When things get really hot, the energy distribution tries to even out, following the spirit of the equipartition theorem. At high T, the average energy ends up being nearly zero because the “ups” and “downs” cancel each other out in the chaos.
Conclusion
Getting a handle on the equipartition of energy is a major win for your IIT JAM 2027 prep. It’s the secret sauce that explains why different gases heat up at different rates. Once you get used to counting degrees of freedom, you’ll start seeing these problems as easy points on the board.
Just remember: the “law” isn’t perfect. When things get super cold, quantum mechanics steps in and ruins the party (we call this the “freezing” of degrees of freedom). But for most of your exam prep, just remember the “equal split” rule and you’ll be golden.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
What is a "Degree of Freedom" (f) in thermodynamics?
It refers to the number of independent coordinates or ways a molecule can move (translate, rotate, or vibrate) in space.
Who proposed the Equipartition Theorem?
The theorem was primarily developed by James Clerk Maxwell and later refined by Ludwig Boltzmann.
Is the equipartition of energy applicable to all systems?
No, it is strictly applicable to classical systems in thermal equilibrium where the energy depends quadratically on the coordinates/momenta.
How many degrees of freedom does a monatomic gas have?
A monatomic gas (like Helium) has 3 degrees of freedom, all of which are translational (f = 3).
What are the degrees of freedom for a diatomic molecule at room temperature?
It typically has 5 degrees of freedom: 3 translational and 2 rotational (f = 5).
Why are vibrational degrees of freedom ignored at room temperature?
Vibrational modes require higher energy levels to be "excited." At room temperature, kB T is usually insufficient to activate these modes.
How many degrees of freedom does a non-linear polyatomic molecule have?
It has 6 degrees of freedom: 3 translational and 3 rotational.
What is the formula for the total internal energy (U) using this theorem?
The formula is U = (f/2) nRT$, where n is the number of moles and $f$ is the degrees of freedom.
Why does the equipartition theorem fail at very low temperatures?
At low temperatures, quantum effects become dominant, and energy levels become discrete (quantized), leading to "frozen" degrees of freedom.
What is the difference between Equipartition and Conservation of Energy?
Equipartition describes the distribution of energy at equilibrium, while Conservation states that total energy remains constant in an isolated system.
What is the most common mistake students make in JAM questions on this topic?
Students often forget to check if the molecule is linear or non-linear before assigning rotational degrees of freedom.
Does the equipartition theorem apply to potential energy?
Yes, but only if the potential energy is a quadratic function of the coordinates (like a simple harmonic oscillator).
How does temperature affect the degrees of freedom?
As temperature increases, more degrees of freedom (like vibration) "unlock" or contribute to the specific heat.














