Conductance is a measure of the ability of a material to conduct electric current. In the context of IIT JAM, students need to understand the fundamental principles of conductivity and its applications in various fields, including electronics and physics.
Electrical Conductance Syllabus and Key Textbooks
Let’s face it: when you first look at the physical chemistry syllabus for IIT JAM, the electrochemistry section can feel a bit overwhelming. But if you break it down, Conductivity is one of those high-yield topics that can seriously boost your score if you get the fundamentals right. In the official syllabus, this sits comfortably within the electrochemistry and basic physical measurement topics.
If you want to move past just memorizing formulas and actually understand what’s happening in the solution, you need the right books. Here are a couple of solid recommendations that we often talk about at VedPrep to get you started:
-
P.W. Atkins – Physical Chemistry: While some Indian entrance guides focus purely on formulas, Atkins helps you visualize how ions actually move through a solvent. It’s perfect for getting a grip on molar and equivalent conductivity.
-
K.L. Kapoor – A Textbook of Physical Chemistry (Vol. 5): This is an absolute gold standard for Indian competitive exams. It lays out the mathematical derivations and numerical problems exactly the way you’ll encounter them in the exam hall.
Conductance and its applications For IIT JAM
At its core, Conductance is just a way of measuring how easily electricity flows through a system. If resistance is the barrier putting up a fight against the current, conductance is the open green light. Mathematically, it’s just the flip side of the coin:
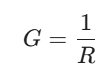
While resistance is measured in Ohms (Ω), we measure conductance in Siemens (S) or ohm-1 (which people fondly call mho).
In the context of the IIT JAM exam, you aren’t just looking at solid copper wires; you are mostly dealing with electrolytic solutions. Think about it this way: how fast can ions move from point A to point B when an electric field is turned on? Understanding this simple concept helps you unlock trickier topics down the road, like predicting circuit behaviors in your basic electronics physics modules or calculating cell potentials.
Common Misconceptions about Conductance and Resistance
It is incredibly common to see students treat conductance and resistance as interchangeable terms during a high-stress exam, but they point in completely opposite directions. Resistance is the traffic jam; conductance is the empty highway.
Another trap people fall into is confusing conductance with the actual electric current. Think of it like a water pipe:
Imagine you have a fictional pipe setup in a backyard. The current is the actual volume of water rushing through every second. The conductance is how wide and clear that pipe is. A wide, clean pipe allows water to flow effortlessly, but it doesn’t mean water is automatically flowing unless you turn the tap on (which is your voltage).
- Conductance is the reciprocal of resistance: G = 1/R.
- Conductivity depends on the material’s properties and its geometry.
Conductance and its applications For IIT JAM
While chemistry students look at ions in a beaker, physics and electronics aspirants look at how electrons behave in semi-conductors. Conductivity plays a massive role in how everyday components like diodes and transistors function.
Take a standard diode, for example. When you apply a forward bias, the internal barrier drops, the conductance shoots up, and current flows easily. Flip the polarity to reverse bias, and the conductance drops to almost zero, blocking the current entirely. This simple on-off behavior is exactly what allows gadgets to rectify AC to DC or regulate voltage.
In the IIT JAM paper, you will often find questions asking you to calculate the small-signal conductance of a transistor or analyze a diode’s current-voltage graph. Outside the exam hall, this principle is the backbone of:
-
The amplifiers boosting the sound in your earphones.
-
Environmental sensors that track changes in moisture or temperature by reading shifts in electrical flow.
-
Industrial grids managing massive power distribution lines.
Exam Strategy: How to Approach Conductance Questions in IIT JAM
When you’re staring down a conductivity question on exam day, you need a structured game plan. The math itself isn’t usually terrifying, but the units will trip you up if you aren’t careful.
Start by cementing the core formula in your head:
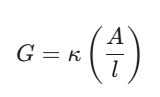
Here, $\kappa$ (kappa) is the specific conductivity, A is the cross-sectional area of the electrodes, and $l$ is the distance between them.
When we analyze student performance data at VedPrep , we notice that the biggest point drops don’t come from a lack of conceptual understanding. They happen because students forget to convert centimeters to meters, or they mix up the definitions of:
-
Specific Conductance (κ): Conductance of a $1\text{ cm}^3$ cube of solution.
-
Molar Conductance (Λm): Conductance behavior when you dissolve exactly one mole of electrolyte.
-
Equivalent Conductance (Λeq): Conductance relative to chemical equivalents, which factors in the valency of the ions.
Keep a close eye on how concentration and temperature change these values. For example, weak electrolytes behave completely differently than strong electrolytes when you dilute them. Sketching out those classic Kohlrausch law graphs during your study sessions will save you a lot of headache later.
By following a structured study plan and leveraging VedPrep’s resources, students can develop a deep understanding of conductance and its applications, ultimately boosting their confidence and performance in the IIT JAM exam.
Real-World Applications of Conductance in Physics
Away from textbooks and exam papers, measuring Conductance is an incredibly powerful tool in modern science and industry.
In materials science, researchers measure how a new alloy or polymer conducts electricity under extreme cold or high pressure to see if it can be used in next-generation batteries. Over in geophysics, scientists use massive electromagnetic sensors to map the earth’s crust. Because salt water, metals, and solid granite all have drastically different electrical conductivities, reading the ground’s conductance helps geophysicists pinpoint underground water reserves, mineral deposits, or even track volcanic activity.
You even see this in healthcare. Medical imaging tools and diagnostic tests rely heavily on tracking how electrical currents pass through different tissues in the human body. Whether it is oil exploration or aerospace engineering, knowing how a material handles electrical flow is vital.
Final Thoughts
As you gear up for the IIT JAM 2027 exam cycle, remember that mastering Conductance isn’t about memorizing a handful of definitions the night before. It is about understanding the physical reality of how particles move—whether those are electrons in a silicone chip or sodium ions migrating through a solution.
If you can connect the dot between the molecular level and the macroscopic formulas, the questions on exam day will feel much more natural. Keep practicing your numerical problems, watch your unit conversions, and break the chapters down into bite-sized pieces.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
What is the difference between conductance and conductivity?
Conductance refers to the ability of a solution to conduct electricity, while conductivity is a measure of the conductance of a solution per unit length. Conductivity is an intensive property, whereas conductance is an extensive property.
What are the types of conductance?
There are two types of conductance: specific conductance (or conductivity) and molar conductance. Specific conductance is the conductance of a solution per unit volume, while molar conductance is the conductance of a solution containing one mole of an electrolyte.
How is conductance measured?
Conductance is measured using a conductivity cell, which consists of two electrodes separated by a known distance. The conductance is calculated by measuring the resistance of the solution between the electrodes and using Ohm's law.
What are the factors that affect conductance?
Conductance is affected by several factors, including the concentration of ions, temperature, and the presence of other electrolytes. Increasing the concentration of ions or temperature generally increases conductance.
What is the relationship between conductance and electrochemistry?
Conductance is a fundamental concept in electrochemistry, as it helps understand the behavior of electrolytes in solution. Electrochemical reactions involve the transfer of ions, which is directly related to conductance.
How is conductance applied in IIT JAM Physical Chemistry?
Conductance is an important topic in IIT JAM Physical Chemistry, with questions often asked on the principles and applications of conductance, including calculations of conductance and conductivity.
What are some common problems related to conductance in IIT JAM?
Common problems related to conductance in IIT JAM include calculating conductance and conductivity, understanding the factors that affect conductance, and applying conductance to electrochemical reactions.
What are some common mistakes made when calculating conductance?
Common mistakes when calculating conductance include incorrect units, not considering the temperature dependence of conductance, and not accounting for the presence of other electrolytes.
How can I avoid confusing conductance and conductivity?
To avoid confusing conductance and conductivity, remember that conductance refers to the ability of a solution to conduct electricity, while conductivity is a measure of conductance per unit length.
What are some common misconceptions about conductance?
Common misconceptions about conductance include thinking that conductance is a measure of the amount of electricity conducted, rather than the ability to conduct electricity.
What is the relationship between conductance and ionic strength?
Conductance is related to ionic strength, as increasing ionic strength can increase conductance by increasing the concentration of ions in solution.
How does conductance relate to electrode kinetics?
Conductance plays a crucial role in electrode kinetics, as it affects the rate of electrochemical reactions by influencing the transport of ions to and from the electrode surface.
How can conductance be used to study complex electrochemical systems?
Conductance can be used to study complex electrochemical systems by providing insights into the behavior of ions and electrochemical reactions in these systems.
What are some future directions for research on conductance in electrochemistry?
Future directions for research on conductance in electrochemistry include the development of new experimental techniques and theoretical models to better understand conductance and its applications.










