Mean free path The mean free path for an IIT JAM is the average distance traveled by a gas molecule between collisions, which is inversely proportional to gas density and critical in understanding kinetic theory and collision dynamics.
Syllabus: Thermodynamics and Statistical Mechanics (IIT JAM)
In the IIT JAM syllabus, this topic sits comfortably inside the thermodynamics and statistical mechanics unit. If you’re also eyeing CSIR NET or GATE down the line, you’ll find this concept follows you there too.
To really get under the hood of this topic, most of us at VedPrep suggest sticking to the classics. Pathria is the gold standard for the statistical mechanics side of things, while Atkins does a great job explaining the physical chemistry perspective. These books help you see the mean free path not just as a variable in a formula but as the reason behind why gases behave the way they do.
Two key textbooks that cover this topic are the following:
- Statistical Mechanics by R K Pathria on Mean Path Length For IIT JAM
- Thermodynamics by P W Atkins on Mean Path Length For IIT JAM
Mean Free Path For IIT JAM: Definition and Importance
Imagine you’re trying to walk through a crowded Delhi metro station during rush hour. You take a few steps, dodge someone, walk another meter, and then almost bump into someone else. The average distance you walk between those “near-misses” is essentially your mean free path.
In a gas, molecules are constantly zipping around. They aren’t just moving in straight lines forever; they’re constantly bouncing off each other like billiard balls. Because these collisions are elastic, they don’t lose energy, but they do change direction. The average of all those straight-line distances between hits is the mean free path. Understanding this is the secret sauce for figuring out things like how fast a smell spreads across a room (diffusion) or how thick a gas feels (viscosity).
Derivation of Mean Free Path For IIT JAM
To derive this for the JAM exam, we usually use the “hard-sphere” model. We pretend molecules are like tiny, hard marbles with a diameter d.
The logic goes like this: if a molecule is moving, it sweeps out a sort of “collision tube.” Any other molecule whose center falls within that tube is going to get hit. When you crunch the numbers and account for the fact that all the molecules are moving (not just one), you get the famous formula:
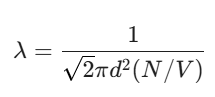
Here, N/V is just the number density (how many molecules are packed into a certain volume). If you cram more molecules in (higher density), the distance they can travel before a hit drops. It’s common sense, really—a crowded room means more bumping.
Worked Example: Mean Free Path for IIT JAM
Let’s look at a typical problem you might see in a mock test.
The Task: Find the mean free path for a gas at 273 K and 1 atm, given a molecular diameter of 2.5 Å.
The Strategy: We use the version of the formula that plugs in pressure (P) and temperature (T):
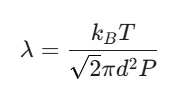
-
Convert everything to SI units: T = 273K, P = 1.013 × 105Pa, and d = 2.5 × 10⁻¹⁰ m.
-
Constants: kB (Boltzmann constant) is roughly 1.38 × 10-23 J/K.
-
Calculate: When you plug those in, you’ll find $\lambda$ is about 1.06 × 10⁻⁷ m.
That might seem small, but for a tiny molecule, that’s hundreds of times its own body length!
Common Misconceptions: Mean Free Path For IIT JAM
A big mistake people make is thinking the mean free path is a fixed, physical “gap” between molecules. It’s not. It’s a statistical average. Some molecules might get lucky and fly across the whole container, while others might get smacked twice in a nanosecond.
Also, don’t get trapped thinking collisions are always like hard marbles. In reality, molecules have attractive and repulsive forces (Van der Waals, anyone?), but for the JAM level, the hard-sphere approximation usually gets the job done.
Real-World Applications: Mean Free Path in Engineering
This isn’t just for passing exams. If you ever move into research or chemical engineering, the mean free path is a dealbreaker.
Take vacuum systems, for example. If you’re trying to coat a lens with a thin film, you need the mean free path to be larger than the size of the chamber. Why? Because you want the atoms to fly straight to the lens without hitting a stray air molecule and getting knocked off course. It’s also why your thermos stays cold—by removing air, you increase the mean free path so much that molecules rarely hit each other to transfer heat.
In engineering, the mean path length for IIT JAM is used to optimize system efficiency and performance. For instance, in vacuum systems, knowledge of Mean Path Length for IIT JAM helps designers create systems with minimal gas collisions, ensuring efficient operation. Similarly, in gas chromatography, understanding the mean free path for IIT JAM is vital for separating and analyzing mixtures of gases, all of which rely on the mean free path for IIT JAM.
- In chemical reactors, mean path length for IIT JAM influences reaction rates and yields.
- In heat transfer systems, it affects energy exchange between gases, both important for mean path length for IIT JAM.
Exam Strategy: Studying Mean Free Path For IIT JAM
When you’re sitting down with your notes, don’t just stare at the formula. Ask yourself: “What happens if I double the pressure?” (The path gets cut in half). “What if the molecules are twice as big?” (The path shrinks by a factor of four because of the $d^2$ term).
Focus on:
-
How λ relates to temperature and pressure.
-
The difference between collision frequency and mean free path.
-
How it connects to the transport properties like thermal conductivity.
Additional Tips: VedPrep’s Resources and Study Materials. Mean free path For IIT JAM
We know that staring at a textbook for four hours can turn your brain to mush. That’s why at VedPrep , we try to break these down into bite-sized pieces. If you’re feeling stuck on the derivation, check out some of our free video lectures. Sometimes seeing someone draw the “collision cylinder” on a whiteboard makes it click way faster than reading a wall of text. We also have quizzes that focus specifically on these KTG concepts so you can test your speed for the NAT section.
Conclusion
Mastering the mean free path is about visualizing the microscopic chaos of a gas. If you can imagine those tiny molecules zipping and bumping, the math starts to feel a lot more natural. Keep your practice consistent; use the resources available at VedPrep when you hit a wall, and you’ll find that these marks are some of the easiest to grab on exam day.
To know more from our faculty, watch our YouTube video:
Frequently Asked Questions
Is the Mean Path Length for IIT JAM a constant value?
No, it is not a fixed value. It is a statistical average that changes based on the gas's environment, such as its temperature, pressure, and density.
Why is the Mean free path for IIT JAM important in thermodynamics?
It is critical for understanding transport phenomena, including thermal conductivity, viscosity, and diffusion, which are major topics in the IIT JAM syllabus.
How does gas density affect the Mean Path Length for IIT JAM?
The Mean free path is inversely proportional to density (n = N/V). As density increases (more molecules in a smaller space), the distance between collisions decreases.
What is the relation between pressure and Mean free path for IIT JAM?
At a constant temperature, the mean free path is inversely proportional to pressure (P). Higher pressure results in a shorter mean path length.
How does temperature influence the Mean free path for IIT JAM?
At constant volume, temperature doesn't change λ. However, at constant pressure, λ is directly proportional to temperature (T), as increasing T causes gas expansion and lower density.
Is Mean Path Length for IIT JAM part of the official syllabus?
Yes, it falls under the Thermodynamics and Statistical Mechanics unit of the IIT JAM Physics and Chemistry syllabi.
What kind of NAT questions are asked on Mean free path for IIT JAM?
Students are often asked to calculate λ given values for P, T, and d, or to find the ratio of path lengths at different temperatures.
Which textbooks are best for studying Mean Path Length for IIT JAM?
Standard references include Statistical Mechanics by R.K. Pathria and Thermodynamics by P.W. Atkins.
How is Mean free path for IIT JAM used in vacuum systems?
In high-vacuum engineering, the mean free path must be larger than the size of the container to ensure "molecular flow" and minimize interference.
Does Mean Path Length for IIT JAM apply to liquids?
While primarily a gas-phase concept, it can be adapted for liquids, though the distance is much shorter due to high density and continuous interaction.
What is the "Mean Free Time" for IIT JAM?
It is the average time interval between two collisions. It is calculated by dividing the mean free path by the average velocity of the molecules.
How does altitude affect the Mean free path for IIT JAM?
As you go higher in the atmosphere, pressure and density decrease significantly, causing the mean free path to increase from nanometers to kilometers in the exosphere.
Are collisions between molecules truly elastic for Mean Path Length for IIT JAM?
For the sake of JAM-level derivations, they are assumed to be perfectly elastic, meaning total kinetic energy is conserved.
Why do we use the "Average Distance" instead of the exact distance?
Molecular motion is random; therefore, the distance between any two specific collisions varies. A statistical average is the only way to describe the system predictably.














