Physisorption and chemisorption are two types of adsorption processes that occur between a solid surface and a gas or liquid molecule. Understanding the differences between these two processes is crucial for IIT JAM and other competitive exams.
Syllabus – Thermodynamics and Surface Chemistry for IIT JAM
This topic falls under the official IIT JAM Chemistry syllabus. The unit covers various aspects of surface chemistry, including adsorption, physisorption, and chemisorption.
Students can refer to standard textbooks such as Atkins’ Physical Chemistry and Atkins’ Inorganic Chemistry for in-depth study of surface chemistry and adsorption processes. These textbooks provide comprehensive coverage of thermodynamic and kinetic principles underlying physisorption and chemisorption.
Key concepts in this unit include adsorption isotherms, adsorption kinetics, and the distinction between physisorption and chemisorption. A thorough understanding of these topics is essential for success in IIT JAM and other competitive exams.
Physisorption and Chemisorption: A Comparative Analysis
Adsorption is basically a surface game. Imagine walking into a crowded Delhi Metro train during peak hours—some people just lean lightly against the doors because they have to get off at the next station, while others grab the central pole with a death grip because they are traveling all the way to the end of the line. That is exactly how molecules behave on a solid surface.
In the world of surface chemistry, the surface providing the space is the adsorbent, and the gas or liquid molecules sticking to it are the adsorbate. Depending on Physisorption and chemisorption, we split the process into physisorption and chemisorption.
Physisorption relies on weak van der Waals forces. Because these interactions are weak and non-specific, the whole process is reversible. It usually happens at low temperatures. If you increase the temperature or drop the pressure, the molecules just pop right off the surface—much like that commuter letting go of the metro door.
As per Physisorption and chemisorption, chemisorption is a whole different story. Here, the adsorbate shares or exchanges electrons with the surface, forming actual, strong chemical bonds. This creates a brand-new chemical surface layer, needs higher temperatures to kick off, and is generally irreversible. Once that bond forms, you cannot easily undo it.
Here is a quick look at how they stack up side by side:
| Feature | Physisorption | Chemisorption |
| Type of Forces | Weak van der Waals forces | Strong chemical bonds |
| Reversibility | Highly reversible | Generally irreversible |
| Temperature | Favored at low temperatures | Favored at high temperatures |
| Specificity | Non-specific (any gas can adsorb) | Highly specific (occurs only if bonds can form) |
| Layers | Multilayered | Monolayered only |
| Activation Energy | Almost zero | Often requires high activation energy |
Physisorption and Chemisorption For IIT JAM: Worked Example
Let’s look at a typical problem you might face in the physical chemistry section of the exam.
Imagine a fictional lab scenario where we expose a clean solid surface to an ideal gas inside a closed container at 300 K. Let’s say we start with $1\text{ mole}$ of this gas, and we want to see how it covers the surface using the Langmuir adsorption isotherm.
If the system reaches equilibrium and we find that exactly 0.5 mole of the gas has stuck to the surface, we can calculate the adsorption coefficient (b).
The Langmuir isotherm equation is:
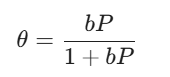
Where:
θ is the fraction of the surface covered.
P is the equilibrium pressure of the gas.
b is the adsorption coefficient.
First, let’s find θ. Since half of our total gas sample is on the surface, the fraction of surface coverage is:
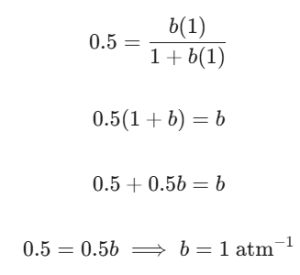
Physisorption and Chemisorption For IIT JAM: Common Misconceptions
When you are deep into your exam prep, it is incredibly easy to mix up the fine print. One big trap students fall into is thinking that physisorption creates a permanent attachment. Because Physisorption and chemisorption relies on weak van der Waals forces, the molecules are constantly hopping on and off. It is a dynamic equilibrium, not a permanent retirement home for gases.
Another common slip-up is assuming chemisorption is always an irreversible one-way street. While it is true most of the time because breaking chemical bonds is tough, there are specific chemical systems where chemisorption can reverse if you pump in enough energy or drastically alter the environment.
Also, don’t think a surface can only do one or the other. In a real reactor, physisorption and chemisorption often happen at the same time. A gas molecule might first land on a catalyst surface via weak physisorption at a low temperature, and then, as the system heats up, find the energy to form a hardcore chemical bond, transitioning fully into chemisorption.
Real-World Applications of Physisorption and Chemisorption
To make these concepts stick, let’s look at how they work outside the textbook.
Think about the catalytic converter in a car. It has to clean up toxic exhaust fumes like carbon monoxide before they exit the tailpipe. Inside, the metal surface (usually platinum or palladium) uses chemisorption to grip the carbon monoxide molecules, breaking their internal bonds so they can react with oxygen to form harmless carbon dioxide. This chemical bonding requires a hot environment, which is why your car converter operates between 300°C and 800°C.
On the flip side, if you have ever used a charcoal water filter or seen industrial gas purification columns, you are looking at physisorption in action. Activated carbon or porous minerals called zeolites act like giant molecular sponges. They don’t form chemical bonds with impurities; instead, they use weak physical attraction to pull contaminants out of the air or water stream. When the filter gets full, you can often heat it up to drive the trapped gases away and reuse the material.
Even biology uses these mechanisms. Suppose a medical implant is placed inside a body. Proteins will instantly blanket the implant’s surface using physisorption, which dictates how the body’s immune cells react to the device. Meanwhile, enzymes in your cells use precise chemical docking—essentially a form of chemisorption—to lock onto substrates and speed up metabolic reactions.
Exam Strategy for Physisorption and Chemisorption For IIT JAM
When you sit down to plan your surface chemistry revision, do not just read the definitions over and over. Focus heavily on graphical questions. You need to know exactly what an adsorption isobar looks like for both processes.
Pro Tip: Remember that a physisorption isobar shows a steady decline in adsorption as temperature goes up. A chemisorption isobar, however, first climbs upward because it needs initial activation energy, before dropping off at even higher temperatures as the bonds begin to stress and break.
Spend some quality time practicing problems that combine the Langmuir and Freundlich isotherms. Work on deriving units for the constants in these equations—examiners love to trip you up there. At VedPrep, we suggest mapping out a quick comparative chart from memory every few weeks to keep the distinct traits fresh in your mind.
Keep an eye out for these high-yield exam subtopics:
The exact thermodynamic criteria (ΔH, ΔS, and ΔG are all negative for spontaneous adsorption).
The effect of pressure variations on the Freundlich exponent (1/n).
How surface area changes influence the total heat of adsorption.
Importance of Physisorption and Chemisorption For IIT JAM
Surface chemistry might seem like a small chapter compared to massive topics like Chemical Kinetics or Coordination Compounds, but it is a regular scoring zone in physical chemistry. Every single mark matters when you are aiming for an IIT or IISc seat.
Understanding how molecules behave on a surface gives you the foundation to solve complex kinetics problems and grasp heterogeneous catalysis, which shows up across both physical and inorganic chemistry papers. If you can confidently explain why physisorption forms multiple layers while chemisorption stops after a single layer of chemical bonds, you are already ahead of a huge chunk of the competition.
Final Thoughts
Nailing the differences between Physisorption and chemisorption is a fantastic way to secure quick marks in your exam. As you map out your study schedule for the IIT JAM 2027 cycle, keep your eyes on the thermodynamic boundaries and practice reading isotherm graphs accurately.
VedPrep offers specialized coaching and curated study modules to help you navigate these complex topics and stay ahead of the competition. With a disciplined approach and clear conceptual clarity, you can turn this high-scoring unit into a major advantage on your journey to a top-tier Indian institute.
To know more in detail from our faculty, watch our YouTube video:
Frequently Asked Questions
Why is physisorption considered a reversible process?
Because the forces involved (van der Waals) are weak, the adsorbed molecules can be easily removed by decreasing pressure or increasing temperature without changing the chemical nature of the substance.
Is chemisorption always irreversible?
Generally, yes, because it involves the formation of strong chemical bonds. However, in specific cases and under high energy/temperature conditions, it can be reversible.
Can physisorption and chemisorption occur simultaneously?
Yes. Often, a gas may initially be physisorbed on a surface and then transition into chemisorption as the temperature increases or as activation energy is met.
Which process has a higher enthalpy of adsorption?
Chemisorption has a much higher enthalpy (80–400 kJ/mol) compared to physisorption (20–40 kJ/mol) due to the strength of the chemical bonds formed.
How does temperature affect physisorption?
Physisorption generally decreases as temperature increases because it is an exothermic process and the weak forces are easily disrupted by thermal energy.
Why does the rate of chemisorption sometimes increase with temperature initially?
Unlike physisorption, chemisorption often requires activation energy to form chemical bonds. Therefore, increasing temperature helps the molecules overcome this energy barrier.
What is the effect of pressure on adsorption?
In both cases, increasing pressure generally increases the amount of gas adsorbed, as described by various adsorption isotherms.
Is "Surface Chemistry" a major part of the IIT JAM syllabus?
Yes, it is a core component of the Physical Chemistry section, specifically within the Thermodynamics and Surface Chemistry unit.
Which textbooks are best for studying Physisorption and Chemisorption?
Atkins' Physical Chemistry and Puri Sharma Pathania are highly recommended for mastering these concepts for competitive exams.
What is the Langmuir Adsorption Isotherm?
It is a mathematical model that describes how the amount of gas adsorbed depends on pressure, assuming monolayer coverage and equivalent adsorption sites.
What are the key assumptions of the Langmuir Isotherm?
The surface is homogeneous, there is no interaction between adsorbed molecules, and only a monolayer (single layer) can be formed.
Which type of adsorption is highly specific?
Chemisorption is highly specific because chemical bonds only form if there is chemical affinity between the adsorbate and the adsorbent.
Why does physisorption form multimolecular layers?
Since it depends on weak universal forces (van der Waals), once one layer is formed, additional layers can sit on top of it through the same weak attractions.
What does the "adsorption coefficient (b)" represent in the Langmuir equation?
It represents the affinity between the gas and the surface. A higher 'b' value means the gas is more strongly adsorbed at a given pressure.










